多光子顕微鏡 Bergamo® IIIシリーズ

Please Wait
多光子顕微鏡Bergamo® IIIシリーズのプラットフォーム
顕微鏡に試料を対応させるのではなく、試料に顕微鏡を対応させるという方針に従い、当社では様々な実験要件に対応する完全モジュール式の多光子イメージングプラットフォームを開発しました。多光子イメージングシステムBergamo IIIシリーズでは、微小電極などを用いた従来の研究手法に比べて、ニューロン集団に対する同時読み出しと操作を、高速、高深度、高信号強度で行うことが可能です。
実験に適した顕微鏡の選択
多光子顕微鏡プラットフォームBergamo IIIはモジュール設計であるため、それぞれの実験ニーズに適した構成に変更したり、設置後に顕微鏡の機能を調整したりすることが可能です。以下では、Bergamo IIIのモジュール性についての概要をご紹介しています。詳細は「モジュール」タブをご覧ください。 顕微鏡Bergamo IIIが研究室でどのように使用されているかについては、「Applications」タブと「Publications」タブをご覧ください。
顕微鏡の試料への接近
顕微鏡Bergamo IIIを試料に近づけられるように、支持用ボディを3種類ご用意しています。回転ボディでは、顕微鏡Bergamo IIIに5軸の可動軸を持たせ、in vivoシステムをほぼ全方向から自由に調べることができます。直立ボディでは、対物レンズの下に広い作業空間を持たせ、1軸または3軸の可動軸を装備しています。
- 回転ボディ
- 試料または観察対象物の周囲を最大90°回転可能
- 垂直方向の粗動範囲:127 mm
- XYの精密移動範囲:50.8 mm
- Zの精密移動範囲:25.4 mm
- 対物レンズとともにX、Y、Zの各軸回りに回転
- 直立ボディ
- XYの精密移動範囲:50.8 mm (XYZ構成のみ)
- Zの精密移動範囲:25.4 mm
- 対物レンズからボディまでの距離(Throat Depth/懐深さ):196.5 mm
- 追加可能なZ軸焦点調整機構(2種類)
- 対物レンズ用高速ピエゾスキャナ
- 無振動で高速イメージングの可能な液晶リモートフォーカス
1つのシステムでの複数手法によるイメージング
顕微鏡Bergamo IIIには、最大2つのレーザ走査路のほかに多数のイメージング用補助モジュールを組み込めるため、1つのシステムで複数のイメージング手法を柔軟に切り替えることができます。またモジュール設計のため、実験要件の変化に合わせて顕微鏡の再構成やアップグレードが可能です。
- シングルまたはデュアル走査路
- 共焦点イメージング
- 同時多光子イメージングと空間光変調
- 透過光イメージング
- Dodt勾配コントラスト(ワイドフィールド、レーザ走査)
- DIC(ワイドフィールド、レーザ走査)
- 可視(VIS)および近赤外(NIR)のLED
- ベッセルビームイメージング

Click to Enlarge
Figure 1.1 こちらの顕微鏡 Bergamo IIIは、多光子イメージングと透過光イメージングの両方用に構成されています。透過照明モジュールWFA1000は簡単に取り外すことができ、生きた試料の多光子イメージングを行うためのスペースを確保できます。
より高速でのデータ取得
顕微鏡Bergamo IIIには、8 kHzおよび12 kHzで走査できるガルバノ-レゾナント(GR)スキャナとレゾナント-ガルバノ-ガルバノ (RGG)スキャナをご用意しており、高速での画像取得が可能です。 高速イメージングと試料の光操作・光活性化を同時に行うために、ガルバノ-ガルバノ(GG)スキャナまたは空間光変調器(SLM)を第2の走査路としてセットアップすることができます。
- 高速イメージング用のGRスキャナとRGGスキャナ (8 kHz、12 kHz)
- ユーザが定義した関心領域(ROI)や光刺激パターンを走査するためのGGスキャナ
- 多点同時刺激用の空間光変調器(SLM)

Click to Enlarge
Figure 1.2 こちらの顕微鏡 Bergamo IIIの主走査路には、高速で画像取得を行うためにガルバノ- レゾナントスキャナが取り付けられています。特定の領域を同時光活性化するために、第2走査路にはガルバノ- ガルバノスキャナが取り付けられています。
試料のより広い範囲の観察
顕微鏡Bergamo IIIは視野数(FN)40で構成することができるため、1つの視野(FOV)内で複数の関心領域(ROI)のイメージングができます。視野を広く構成したときには、視野の一部の小さな部分を操作するための第2の走査路を追加できます。
- FN40、8 kHzスキャナ、1倍ズームで取得したときのイメージのサイズ
- FOV:2.82 mm x 2.82 mm(倍率10倍の対物レンズ使用時)
- FOV:1.88 mm x 1.88 mm(倍率15倍の対物レンズ使用時)
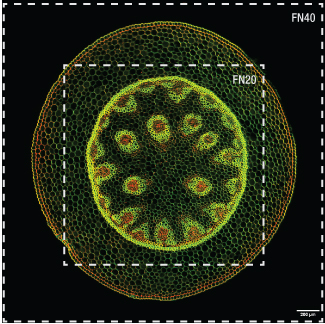
Click to Enlarge
Figure 1.3 上の画像は、視野数(FN) 40で構成された顕微鏡Bergamo IIIに、
当社の対物レンズTL10X-2Pを取り付けて取得しました。
外側の枠がFN40による視野2.82 mm x 2.82 mmの範囲です。
高感度検出による試料の保護
検出効率を最大化するため、顕微鏡Bergamo IIIでは高感度GaAsP PMTをノンデスキャン検出器として使用しています。こうすることで、試料深部からの微弱な信号を検出したり、試料を光損傷させないためにレーザ光パワーを低くして測定したりすることができます。
- 最大で4つの同時検出チャンネルを使用可能
- ノンデスキャン検出器
- 高感度GaAsP PMT
- 微弱信号用の冷却PMT
- より広い集光角度を有する非冷却PMT
- マルチアルカリPMTもご用意
- ほかのイメージング手法のための自由空間光用フォトディテクタ
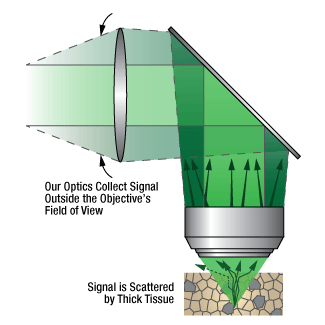
Click to Enlarge
Figure 1.4 信号光が試料から放出されるときに厚い組織によって散乱されるため、当社では広い集光角度を有する検出モジュールを設計しました。8°、10°または14°の集光角度(Ø20 mmの入射瞳の場合)を有する当社独自の検出モジュールを使用すると、深い組織からの生理学的なイメージングが可能になります。
スーパー広帯域走査光学系による深部の観察
顕微鏡Bergamo IIIは、最新のレーザや蛍光色素、さらに最新の技術にも対応できる励起波長範囲をカバーしています。走査光学系は1800 nmまでの波長に最適化することができ、3光子イメージング技術を用いて試料の奥深くまで観察することができます。
- 下記の用途に最適化されたスーパー広帯域走査光学系
- 光刺激/光照射アンケージング
- 2光子イメージング
- 3光子イメージング
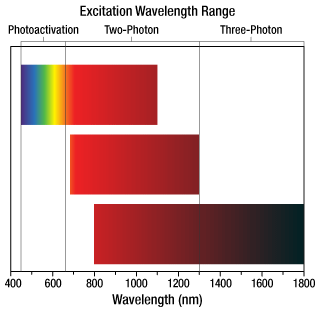
Click to Enlarge
Figure 1.5 当社のスーパー広帯域走査光学系は、光刺激、2光子、3光子でイメージングするための波長範囲に対応しています。
超安定の多光子顕微鏡プラットフォーム
すべてのBergamo III顕微鏡のボディには、走査路のうちの一方または両方に、ファイバ結合型の2光子レーザを組み込むことができます。ファイバ結合により複雑なアライメント手順が不要になり、自由空間レーザ光を使用するよりも安定したイメージングシステムを実現できます。
- イメージングのためのアライメントが不要
- 安定性の高いイメージングシステム構成
- 顕微鏡の設置面積の小型化により、試料や観察対象物のためのスペースを確保
- 顕微鏡の再構成を迅速かつ容易に実施可能

Click to Enlarge
Figure 1.6 このBergamo顕微鏡ではファイバ結合のデュアル走査路を使用しています。
顕微鏡Bergamo® IIIのモジュール
顕微鏡Bergamo IIIは、実験の要件に合わせ構成をカスタマイズできるモジュール式のシステムです。

Click to Enlarge
Figure 2.1 こちらの顕微鏡 Bergamo IIIの主走査路には、高速で画像取得を行うためにガルバノ- レゾナントスキャナが取り付けられています。特定の領域を同時光活性化するために、第2走査路にはガルバノ- ガルバノスキャナが取り付けられています。
ガルバノ-レゾナントスキャナ、ガルバノ-ガルバノスキャナ、空間光変調器(SLM)
顕微鏡Bergamo IIIは、入射ビームの伝搬、調整、およびその方向付けをするための、事前に定めた1つまたは2つの走査路で構成することができます。各走査路には、レゾナント-ガルバノ-ガルバノスキャナ、ガルバノ-レゾナントスキャナ、ガルバノ-ガルバノスキャナ、または空間変調器(SLM)をご使用いただけます。これらから選択することで、高フレームレート、高感度、さらには関心領域(ROI)へのターゲット照射など、各実験に必要な条件に合わせて走査系を最適化できます。
マルチモーダル走査に適したレゾナント-ガルバノ-ガルバノスキャナ
当社では8 kHzおよび12 kHzのレゾナント-ガルバノ-ガルバノスキャナ(RGG)をご用意しています。当社のRGGスキャナの設計は特許取得済みです(US Patent 10,722,977)。このマルチモーダルスキャナのスキャンヘッドは1つですが、ガルバノ-レゾナントスキャナとガルバノ-ガルバノスキャナの両方の機能が付いています。8 kHzスキャナは視野全体を使用し、最大フレームレートは400 fpsです。一方、12 kHzスキャナの最大フレームレートは600 fpsです。
高速イメージングに適したガルバノ-レゾナントスキャナ
当社では、8 kHzと12 kHzのガルバノ-レゾナントスキャナをご用意しています。8 kHzスキャナは視野全体を使用し、最大フレームレートは400 fpsです。一方、12 kHzスキャナの最大フレームレートは600 fpsです。
ユーザ指定の関心領域(ROI)の形状を走査するのに適したガルバノ-ガルバノスキャナ
ガルバノ-ガルバノスキャナは、お客様が描いた形状(線、ポリライン、正方形、長方形)での走査のほか、光刺激のためのカスタムパターン(円、楕円、多角形、点)での走査もサポートしています。信号の積分時間と画像を均一化するために、ピクセル滞留時間は一定に保たれています。
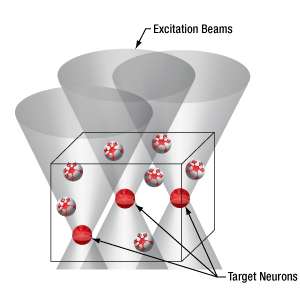
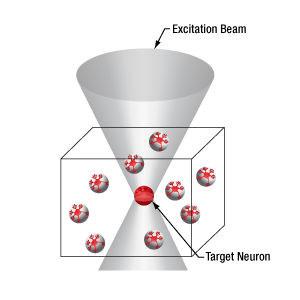
多点同時光刺激に適した空間光変調器(SLM)
物理的に点から点に移動するスキャナとは異なり、空間変光変調器(SLM)はホログラフィ原理に基づいてビームを回折させ、ユーザが定義するパターンを成形します。このパターン成形には一般的なビーム成形のほか、視野内に多数の集光点を生成することも含まれます。この後者のパターンでは、試料内の複数の部位を同時に光励起することができます。空間光変調器についての詳細は「ハイライト」タブをご覧ください

Click to Enlarge
Figure 2.2 対物レンズTL15X-2PのZ方向焦点位置を微調整するために、対物レンズ用ピエゾスキャナPFM450Eが取り付けられた正立顕微鏡Bergamo III
ボリュームイメージングのための高速Z軸焦点調整(オプション)
当社では顕微鏡Bergamo IIIの高速Z軸焦点調整用に、液晶技術をベースにしたリモート焦点調整システムと、ピエゾ素子を用いた対物レンズ用高速スキャナPFM450Eの2種類をご用意しています。
液晶リモート焦点調整
当社のリモート焦点調整システムは、液晶レンズを用いて焦点面の位置を離散した16の位置の間で高速に切り替えます。従って振動が発生せず、焦点位置や試料に影響を与えることがありません。Z方向の移動範囲は対物レンズに依存します。
対物レンズ用ピエゾスキャナ
対物レンズ用ピエゾスキャナは、閉ループ動作で450 µmのZ方向移動範囲を分解能3 nmで走査することができ、最大ステップ幅100 µmまでの整定時間は25 ms(典型値)です。顕微鏡の焦点位置の正確な設定や、高速・高分解能でのZ走査に適しています。

Click to Enlarge
Figure 2.4 ガウシアンビームの拡大画像

Click to Enlarge
Figure 2.3 ベッセルビームの拡大画像
ベッセルビームを用いたボリュームイメージング
顕微鏡Bergamo IIIではベッセルビームを用いて超高速イメージングを行うことができ、神経回路や神経相互作用のビデオレートでのin vivoボリューム機能イメージングを行うことが可能です。この特殊なビームは非回折で自己修復性を有するため、組織を透過する際に集光状態が長く維持され(深い被写界深度の実現)、さらに再形成することも可能です。この技術は当社の多光子顕微鏡Bergamo IIIと2光子メゾスコープでご利用いただけます。
Figure 2.3と2.4はベッセルビームとガウシアンビームの画像です。画像でご覧いただけるように、ガウシアンビームは1つの焦点に集光し、次第に中心から発散して光は弱くなります。一方ベッセルビームでは円環状のビームが形成され、集光状態が維持されます。
この手法についての詳細は、「ハイライト」タブをご覧ください。

Click to Enlarge
Figure 2.5 直立ボディを用いたBergamo IIIシリーズのシステムにはペリスコープが付いているため、顕微鏡のXYZ各軸の全移動範囲で光学性能を損なわずに使用できます。

Click to Enlarge
Figure 2.6 こちらのBergamo IIIの2つの走査路はファイバ結合されており、設置面積がコンパクトでin vivo用に適しています。
ペリスコープとファイバ結合
多光子顕微鏡に用いられるレーザ光のほとんどは自由空間ビームとして供給されます。Bergamo IIIの焦点を中心にして、対物レンズを最大4軸方向(X、Y、 Z、θ)に移動できる機能を有しているため、ビーム光路もアライメントを維持しながら同じ軸に沿って移動することが求められます。Bergamo IIIシリーズのシステムでは、多関節式のペリスコープを用いることでこの課題を克服しています。ペリスコープは、Bergamoの直立ボディと回転ボディの両方で構成することができ、これらはすべての励起波長に対応します。
当社では、Bergamo IIIの走査路のうちの一方または両方を、ファイバ結合にすることも可能です。ファイバ結合型レーザを使用すると、自由空間レーザのような複雑なアライメント手順が不要になり、超安定のイメージングシステムを構成できます。またペリスコープと比較して場所を取りません。 顕微鏡Bergamo IIIのファイバ結合は2光子レーザ用に設計されており、何れの顕微鏡ボディで構成されたシステムにも対応します。

Click to Enlarge
Figure 2.7 吸収フィルタとダイクロイックキューブは、PMT検出モジュールの前面にある磁石で密閉されたドアの背面側に保持されます。
スーパー広帯域走査光学系
Bergamo IIIシリーズの顕微鏡では、450~1100 nm、680~1300 nm、または800~1800 nmの励起波長用に最適化と補正が施された、当社独自の走査用光学系を使用しています。これらの光学系は、それぞれ光刺激、2光子イメージング、3光子イメージングに適しています。可視(VIS)域から近赤外(NIR)域までのこの広い波長範囲は、最新のOPOシステムや2波長出力レーザに対応するように選ばれています。
信号受光角度の広い光学系
検出システムの最も重要な目標は、限られた光子数の信号からからより多くの信号光子を検出することです。PMTを対物レンズの直後に配置すると(「ノンデスキャン」配置)、試料によって散乱された光は、それが対物レンズの視野外で散乱された光でもPMTに入射し、信号として加算されます。これは多光子顕微鏡に特有の利点です。対物レンズの設計視野以外からも集光することで、組織深部をイメージングする際に全体の検出効率が大幅に向上します。
後方(Epi Direction)に配置する場合は集光角度が8°、10°または14° の光学系を、前方(Transmitted Direction)に配置する場合は集光角度が13°の光学系をご用意しています(角度は入射瞳径Ø20 mmの対物レンズを使用した場合)。 集光モジュールは、機械式シャッタを取り付けて光刺激実験に用いることもできます。
アクセスが容易な吸収フィルタとダイクロイックホルダ
顕微鏡システムBergamo IIIシリーズは、Ø25 mmの蛍光フィルタおよび25 mm x 36 mmのダイクロイックミラーが含まれる業界標準の蛍光フィルターセット に対応しています。当社の検出モジュールは、他社設計とは異なり磁石固定式のホルダになっているため、素早く簡単にフィルタを交換して別の測定に移行できます。当社では広域用のØ32 mm蛍光フィルタと32 mm x 42 mmダイクロイックミラーを用いる検出モジュールもご用意しています。それらを用いれば、より大きい集光角度でより多くの信号を検出できます。
後方(Epi Direction)および前方(Transmitted Direction)での検出
当社の多光子顕微鏡システムでは、高感度のGaAsP PMTを採用しています。量子効率が高いため、微弱な蛍光や感光性の高い試料のイメージングに有効です。PMTとしては、微弱な信号の検出感度を向上させる熱電冷却方式と、パッケージサイズを小さくして開口数を大きくできる非冷却方式のどちらでもご使用いただけます。また、マルチアルカリPMTもご用意しています。
Bergamo IIIシリーズのすべての顕微鏡は、後方に2つまたは4つの検出器、および/または前方に2つの検出器を取り付けることが可能です。後方に設定したPMTで検出する蛍光タグを、前方に配置した検出チャンネルでも検出できるため、蛍光強度が微弱な薄い試料に対して顕微鏡の感度を向上させることが可能です。

Click to Enlarge
Figure 2.8 Bergamo III XYZ回転ボディ用コントローラ
タッチパネル付き多軸コントローラ
こちらのコントローラは回転ボディを用いたBergamo IIIシリーズ用に設計されています。ノブを使って、最大5軸までの電動制御ができます。回転式システムでは、対物レンズ焦点の微調整と筐体の昇降によるZ軸移動をロッカースイッチで切り替えます。各軸の移動操作は個別にロックすることができ、筐体の特定方向(軸)の位置を保持することができます。
タッチパネルを用いて2つの位置の保存と読み出しができます。PCのThorImage®LS上では、最大8つの位置を保存可能です。タッチパネルから、すべてのモータの位置の読み出しもできます。
対物レンズ
Bergamo IIIシリーズの顕微鏡には、M34 x 1.0、M32 x 0.75、M25 x 0.75またはRMSネジ付きの無限遠補正対物レンズを取付けられます。これらから選択すれば、多光子顕微鏡に使用される低倍率で高NAの主な対物レンズが得られます。当社でご用意している最大視野数40までの走査用光学系を使用すると、これらの多光子イメージング用の特殊な対物レンズの光学設計を十分に活用することができ、同じ対物レンズを使用した他社製顕微鏡と比べても集光性能が高くなっています。

Click to Enlarge
Figure 2.10 手動の高剛性スタンド
高剛性スタンドサンプルホルダ
当社の高剛性スタンドサンプルホルダには回転や固定が可能な薄型のプラットフォームが付いており、これにはスライド、記録チャンバ、Z軸ピエゾステージなどのほか、カスタム仕様の実験器具なども取り付けられます。取付け部品は受動的に振動を減衰させるためにØ38 mm(Ø1.5インチ)ステンレススチール製ポストで支えられ、そのポストは赤いポストホルダでワークステーションに固定されます。
試料設置用プラットフォームの高さはロック用カラーで保持されており、回転させることで光路上への試料の設置/撤去を簡単に行えます。また位置が決まった後は、クイックリリース機構でポストを固定できます。
電動式の垂直移動型高剛性スタンドMPM250もご用意しており、25.4 mmの移動範囲で精密な高さ調整が可能です。

Click to Enlarge
Figure 2.11 CMOSカメラZeluxとsCMOSカメラQuantulux
サイエンティフィックカメラ
当社の低ノイズのsCMOSおよびCMOSカメラは、当社の多光子顕微鏡システムに完全対応するよう設計されています。ワイドフィールド顕微鏡および蛍光顕微鏡用として有用であり、in vitroやin vivoの試料からの反射光や蛍光を可視化できます。落射蛍光照明モジュールと組み合わせて使用すると、基準マーカの位置を特定するのに役に立ちます。また、この方法はレーザ光照射が不要なイメージング手法にもお使いいただけます。
当社のカメラは、当社が開発したソフトウェアパッケージThorCamで操作できます。sCMOSカメラには2.1 MPセンサが取り付けられています。CMOSカメラでは、1.3 MP、2.3 MP、5 MP、8.9 MPまたは12.3 MPセンサの取り付けられた製品をご用意しています。一般にカメラの解像度が低いと最大フレームレートは高くなります。これらのカメラには、外部トリガ信号によって画像取得を行うための、独立した補助ポートも付いています。
Bergamo IIIシリーズの顕微鏡には、業界標準のCマウント用またはCSマウント用のネジが付いたカメラも直接取り付けられます。

Click to Enlarge
Figure 2.12 対物レンズの下に設置された透過照明モジュール、電動式コンデンサ、および高剛性スタンドサンプルホルダ
お客様ご自身で取付け可能なDodtコントラスト法およびDIC法によるイメージングモジュール
Bergamo IIIシリーズの顕微鏡はモジュール式であるため、in vitroとin vivoでの実験の切り替えが非常に簡単です。Dodtコントラスト法、レーザ走査型Dodtコントラスト法、およびDIC法にご使用いただける透過照明モジュールは、お客様ご自身で本体への取付け・取外しを5分未満で行うことができます。これらのモジュールは、回転ボディと直立ボディの両方にご用意しております。
各製品には3軸コントローラが付属しています。それを用いてコンデンサ用の電動ステージを25.4 mmの範囲で移動させ、照明条件を最適化することができます。この汎用性の高い設計により、Nikon製のコンデンサや高NA油浸コンデンサにも対応できます。
これらのモジュールを補完するために、当社では透過照明モジュールと対物レンズの間にスライドを適切に配置できる薄型の高剛性スタンドサンプルホルダをご用意しています。
当社では、用途ごとのさまざまなご要望にお応えできるように、
お客様のニーズに合わせたご提案を心掛けています。
ご意見・ご要望、またご質問などございましたら当社までお気軽にご連絡ください。
ハイライト
ベッセルビームを用いた高速ボリューム画像取得
当社ではHoward Hughes Medical Institute(HHMI)とNa Ji教授(カリフォルニア大学バークレー校)の協力を得て、Bergamo®多光子レーザ走査型顕微鏡用のベッセルビームモジュールをご提供しています。 神経細胞活動のin vivoボリュームイメージングを行うには、サブミクロンの空間分解能とミリ秒の時間分解能の両方が必要です。従来の方法では、回折限界のガウシアンビームを走査して3次元画像を作成しますが、ベッセルビームを用いた多光子イメージングでは、軸方向に細長い集光スポットを利用してボリューム画像を取得します。励起光の被写界深度が拡大されたことにより3次元の体積が2次元に投影され、2次元フレームレートは効率的に3次元ボリュームレートに変換されます。
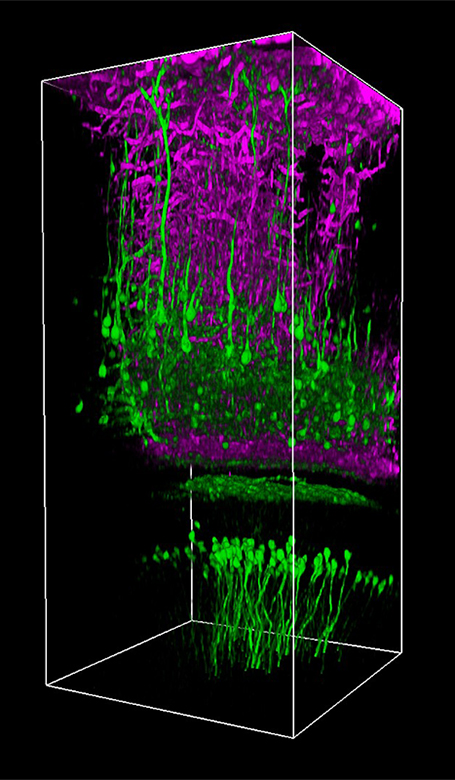
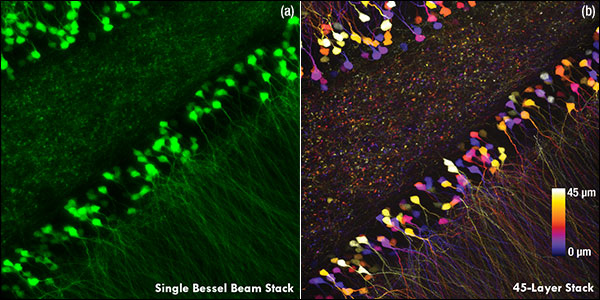
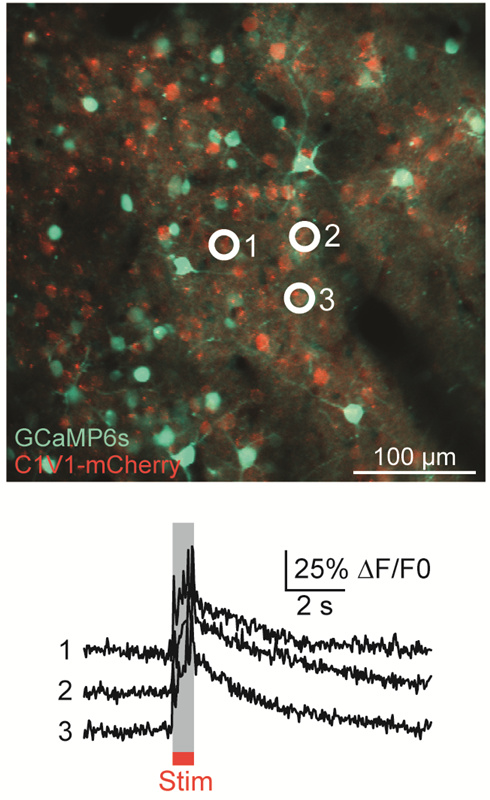
Ji氏の先駆的な研究で実証されたように、このベッセルビームを用いた高速のイメージング技術はシナプス解像度を有し、Ca2+ダイナミクスや、マウスとフェレットの視覚皮質における樹状突起スパインの調整特性などの計測ができます。 Figure 3.1ではこのベッセルビームを用いた多光子イメージング技術の能力を示しています。Thy1-GFP-Mマウスの脳切片の300 x 300 μmの範囲を、それぞれベッセルビーム(Figure 3.1 (a))およびガウシアンビーム(Figure 3.1 (b))で走査した画像を比較しています。ガウシアンビームを集光して取得された45枚の光学的なスライス画像を垂直に積み重ねるとボリュームイメージが得られますが、長さ45 μmの集光スポットを有するベッセルビームを使用すると、1回走査するだけで同じ構造的な特徴を見ることができます。これはボリュームイメージングの速度が大幅に向上することを示しており、この手法はin-vivoでまばらにラベル付けされた試料を調べるのに適しています。
お持ちのBergamo顕微鏡にベッセルビームによるイメージング機能の追加を希望される場合は、当社までご連絡ください。ご提供可能なアップグレードとアドオンについては、「レトロフィット」のタブをご覧ください。
出典: Lu R, Sun W, Liang Y, Kerlin A, Bierfeld J, Seelig JD, Wilson DE, Scholl B, Mohar B, Tanimoto M, Koyama M, Fitzpatrick D, Orger MB, and Ji N. "Video-rate volumetric functional imaging of the brain at synaptic resolution." Nature Neuroscience. 2017 Feb 27; 20: 620-628.
Figure 3.1 1回のベッセルビームによる走査画像(a)は、ガウシアンビームによる45枚の光学画像の断片を積み重ねて得られた体積走査画像 (b)と同じ構造情報を捉えており、これは全走査時間を45分の1に短縮できることを示しています。これらの画像は脳切片の300 μm x 300 μmの範囲を走査したものです。ガウシアンビームによる画像を積み重ねた画像の深さはスケールバーで示されています。試料ご提供:Qinrong Zhang, PhD and Matthew Jacobs; the Ji Lab, Department of Physics, University of California, Berkeley.
Click to Enlarge
Figure 3.3 Thy1-YFPマウス(オス、21週)の画像。1300 nm、繰り返し周波数326 kHz、パルス幅約60 fsで取得。観察窓の中心は、体性感覚皮質上部、ブレグマから横方向に2.5 mm、後方に2 mmの位置にあります(皮質表面でのレーザーパワーは1.1 mW)。画像ご提供:Chris Xu Group, Cornell University.
3光子イメージング
当社では、Bergamo多光子顕微鏡に3光子イメージング技術を導入できるように、800~1800 nmの波長範囲に対応する走査光路用の光学素子を開発しました。3光子励起は深部組織イメージングに適していますが、一般には波長1300 nmまたは1700 nm付近の高パルスエネルギーの励起光源が必要です。2光子イメージングと比較して、3光子イメージングでは組織による散乱が少なく、また焦点の合わないバックグラウンド光が抑制されるので、信号対バックグラウンドの比が改善されます。
Figure 3.2のように、3光子イメージングが可能なシステムの構成要素として、2光子と3光子の同時イメージングをサポートするダイクロイックミラーや、広帯域でのサンプリングができる繰り返し周波数の低いレーザをサポートするエレクトロニクスを含めることができます。ThorImage®LSソフトウェアでは、3光子検出のための重要な機能を強化しています。例えば、3光子信号の検出を励起パルスと同期させ、最良の信号対雑音比が得られるように位相遅延を制御することができます。詳細については「ThorImageLS」タブをご参照ください。
お持ちのBergamo顕微鏡に3光子イメージング機能の追加を希望される場合は、当社までご連絡ください。ご提供可能なアップグレードとアドオンについては、「レトロフィット」タブをご覧ください。
出典: Wang T and Xu C. "Three-photon neuronal imaging in deep mouse brain." Optica. 2020; 7 (8): 947-960.

Click to Enlarge
Figure 3.2 2光子および3光子イメージングの構成。
空間光変調器(SLM)を用いた同時多点光刺激
当社の空間光変調器(SLM)を用いると、ホログラフィックパターンにより試料内の複数の位置の光刺激を同時に行うことができます。この空間光変調器はフェムト秒パルスレーザによる2光子励起用に設計されており、光刺激用レーザのビームプロファイル全体に渡って位相を操作し、ユーザが決定した数百の焦点を生成することができます。
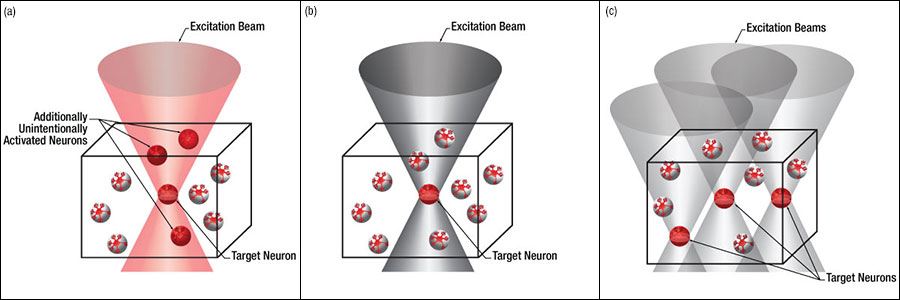
Figure 3.4では、空間光変調器を使用した2光子刺激の利点を、通常の2光子刺激や単一光子による光刺激と比較して示しています。単一光子による光刺激では、1つの細胞だけをターゲットにすることができず、ターゲットとする細胞に近接する細胞も刺激してしまいます。この問題は、1つの細胞をターゲットにできる分解能を有する2光子刺激を利用することで解決します。しかし、その場合でも同時にターゲットにできる細胞は1つに制限されます。2光子刺激と空間光変調器を組み合わせると、多数の焦点を生成して複数の細胞を同時に刺激することができるため、その制限を克服することができます。刺激用ビームの形状は光刺激の効果が向上するように成形することができます。これは、単一視野内の異なる深度にある神経集団を活性化させるために重要な機能です。 空間光変調器の位相マスクのパターンは素早く切り替えることができ、複数の独立した焦点を任意のシーケンスで個々のターゲットに向けることができます。校正プロセス、ホログラムの生成、外部ハードウェアとの同期などは、すべてThorImage®ソフトウェアで管理され、シームレスな制御が可能です。 詳細については「ThorImageLS」タブをご覧ください。
お持ちのBergamo顕微鏡に空間光変調器(SLM)によるイメージング機能の追加を希望される場合は、当社までご連絡ください。ご提供可能なアップグレードとアドオンについては、「レトロフィット」タブをご覧ください。
Figure 3.4 2光子刺激+空間光変調器(c)による方法では、複数のターゲット細胞を同時に刺激することが可能です。これは、単一光子(a)や2光子(b)で刺激する一般的な方法では不可能です。
Thorlabs' Bergamo® III Series Multiphoton Microscopy Platform is a powerful tool adapted to meet experimental needs across a wide range of research fields. Click on the images below to explore how a Bergamo can and has been utilized for each application.
Images Taken Using Bergamo® Systems
Selected Publications Using Thorlabs' Imaging Systems
2025
De Faveri, F., Ceriani, F., & Marcotti, W. (2025). In vivo spontaneous Ca2+ activity in the pre-hearing mammalian cochlea. Nature Communications, 16(1), 29.
Romanova, N., Sule, K., Issler, T., Hebrok, D., Persicke, M., Thévenod, F., Prenner, E. J., & Lee, W. K. (2025). Cadmium-cardiolipin disruption of respirasome assembly and redox balance through mitochondrial membrane rigidification. Journal of Lipid Research, 100750.
Vargas-Ortiz, J., Lin, L., Martinez, V. K., Liu, R.-J., Babij, R., Duan, Z. R. S., ... & Che, A. (2025). Translaminar synchronous neuronal activity is required for columnar synaptic strengthening in the mouse neocortex. Nature Communications, 16(1), 1296.
Das, A., Borovicka, J., Tobin, H., Bhatia, S., Icardi, J., Tahmasebi, G., ... & Dana, H. (2025). Neuronal Silencing and Protection in a Mouse Model of Demyelination. bioRxiv, 2025-02.
Yu, P., Yang, Y., Gozel, O., Oldenburg, I., Dipoppa, M., Rossi, L. F., ... & Doiron, B. (2025). Circuit-Based Understanding of Fine Spatial Scale Clustering of Orientation Tuning in Mouse Visual Cortex. bioRxiv, 2025-02.
Singh, S., Kim, H., Ecevitoglu, A., Chasse, R., Ludko, A. M., Sanganahalli, B., ... & Bae, B.-I. (2025). Autism-associated ASPM variant causes macrocephaly and social-cognitive deficits in mice. bioRxiv, 2025-02.
Goltstein, P. M., Laubender, D., Bonhoeffer, T., & Hübener, M. (2025). A column-like organization for ocular dominance in mouse visual cortex. Nature Communications, 16(1), 1926.
2024
Mittelstadt, J. K., Shilling-Scrivo, K. V., & Kanold, P. O. (2024). Long-term training alters response dynamics in the aging auditory cortex. Hearing Research, 444, 108965.
Braun, J., Hurtak, F., Wang-Chen, S., & Ramdya, P. (2024). Descending networks transform command signals into population motor control. Nature, 630(8017), 686-694.
Chen, W., Ge, X., Zhang, Q., Natan, R. G., Fan, J. L., Scanziani, M., & Ji, N. (2024). High-throughput volumetric mapping of synaptic transmission. Nature Methods, 21(7), 1298-1305.
González-Rueda, A., Jensen, K., Noormandipour, M., de Malmazet, D., Wilson, J., Ciabatti, E., ... & Tripodi, M. (2024). Kinetic features dictate sensorimotor alignment in the superior colliculus. Nature, 631(8020), 378-385.
Dobler, Z., Suresh, A., Chari, T., Mula, S., Tran, A., Buonomano, D. V., & Portera-Cailliau, C. (2024). Adapting and facilitating responses in mouse somatosensory cortex are dynamic and shaped by experience. Current Biology, 34(15), 3506-3521.
Gautham, A. K., Miner, L. E., Franco, M. N., Thornquist, S. C., & Crickmore, M. A. (2024). Dopamine biases decisions by limiting temporal integration. Nature, 632(8026), 850-857.
Xin, W., Kaneko, M., Roth, R. H., Zhang, A., Nocera, S., Ding, J. B., ... & Chan, J. R. (2024). Oligodendrocytes and myelin limit neuronal plasticity in visual cortex. Nature, 633(8031), 856-863.
Shiozaki, H. M., Wang, K., Lillvis, J. L., Xu, M., Dickson, B. J., & Stern, D. L. (2024). Activity of nested neural circuits drives different courtship songs in Drosophila. Nature Neuroscience, 27(10), 1954-1965.
Martineau, É., Malescot, A., Elmkinssi, N., & Rungta, R. L. (2024). Distal activity patterns shape the spatial specificity of neurovascular coupling. Nature Neuroscience, 27(11), 2101-2114.
Kazanovich, I., Itzhak, S., & Resnik, J. (2024). Experience-driven development of decision-related representations in the auditory cortex. EMBO reports, 26(1), 84-100.
Wagenmaker, A., Mi, L., Rozsa, M., Bull, M. S., Svoboda, K., Daie, K., ... & Jamieson, K. G. (2024). Active learning of neural population dynamics using two-photon holographic optogenetics. Advances in Neural Information Processing Systems, 37, 31659-31687.
Fuhrmann, F., Nebeling, F. C., Musacchio, F., Mittag, M., Poll, S., Müller, M., ... & Fuhrmann, M. (2024). Three-photon in vivo imaging of neurons and glia in the medial prefrontal cortex with sub-cellular resolution. bioRxiv, 2024-08.
Zhu, Y., Gelnaw, H., Auer, F., Hamling, K. R., Ehrlich, D. E., & Schoppik, D. (2024). Evolutionarily conserved brainstem architecture enables gravity-guided vertical navigation. PLoS Biology, 22(11), e3002902.
Morton, R. A., & Kim, T. N. (2024). Viscocohesive hyaluronan gel enhances stability of intravital multiphoton imaging with subcellular resolution. Neurophotonics, 12(S1), S14602-S14602.
Kritsilis, M., Vanherle, L., Rosenholm, M., In't Zandt, R., Yao, Y., Swanberg, K. M., ... & Lundgaard, I. (2024). Loss of glymphatic homeostasis in heart failure. Brain, awae411.
Ivan, V. E., Tomàs-Cuesta, D. P., Esteves, I. M., Luczak, A., Mohajerani, M., McNaughton, B. L., & Gruber, A. J. (2024). Psilocybin reduces functional correlation and the encoding of spatial information by neurons in mouse retrosplenial cortex. European Journal of Neuroscience, 60(10), 6395-6407.
Sheahan, T. D., Warwick, C. A., Cui, A. Y., Baranger, D. A., Perry, V. J., Smith, K. M., ... & Ross, S. E. (2024). Kappa opioids inhibit spinal output neurons to suppress itch. Science Advances, 10(39), eadp6038.
Lemeshko, P., Korepanov, O., Podkovyrina, E., Spivak, Y., Moshnikov, V., & Kozodaev, D. (2024). Porous silicon photoluminescence enhancement by silver dendrites registered with multiphoton microscopy. Optics & Laser Technology, 181, 111825.
Farrants, H., Shuai, Y., Lemon, W. C., Monroy Hernandez, C., Zhang, D., Yang, S., ... & Schreiter, E. R. (2024). A modular chemigenetic calcium indicator for multiplexed in vivo functional imaging. Nature Methods, 1-10.
Bowen, Z., De Zoysa, D., Shilling-Scrivo, K., Aghayee, S., Di Salvo, G., Smirnov, A., ... & Losert, W. (2024). NeuroART: Real-time analysis and targeting of neuronal population activity during calcium imaging for informed closed loop experiments. eNeuro.
Ou, Z., Duh, Y. S., Rommelfanger, N. J., Keck, C. H., Jiang, S., Brinson Jr, K., ... & Hong, G. (2024). Achieving optical transparency in live animals with absorbing molecules. Science, 385(6713), eadm6869.
Kopyeva, I., Goldner, E. C., Hoye, J. W., Yang, S., Regier, M. C., Bradford, J. C., ... & DeForest, C. A. (2024). Stepwise Stiffening/Softening of and Cell Recovery from Reversibly Formulated Hydrogel Interpenetrating Networks. Advanced Materials, 2404880.
Ferron, L., Harding, E. K., Gandini, M. A., Brideau, C., Stys, P. K., & Zamponi, G. W. (2024). Functional remodeling of presynaptic voltage-gated calcium channels in superficial layers of the dorsal horn during neuropathic pain. Iscience, 27(6).
Carlton, A. J., Jeng, J. Y., Grandi, F. C., De Faveri, F., Amariutei, A. E., De Tomasi, L., ... & Marcotti, W. (2024). BAI1 localizes AMPA receptors at the cochlear afferent post-synaptic density and is essential for hearing. Cell reports, 43(4).
Westeinde, E. A., Kellogg, E., Dawson, P. M., Lu, J., Hamburg, L., Midler, B., ... & Wilson, R. I. (2024). Transforming a head direction signal into a goal-oriented steering command. Nature, 626(8000), 819-826.
Batalov, I., Filteau, J. R., Francis, R. M., Jaindl, G., Orr, L., Rapp, T. L., ... & DeForest, C. A. (2024). Grayscale 4D Biomaterial Customization at High Resolution and Scale. bioRxiv, 2024-01.
Brockett, A. T., & Francis, N. A. (2024). Psilocybin biphasically modulates cortical and behavioral activity in mice. bioRxiv, 2024-01.
Goldberg, A. R., Dovas, A., Torres, D., Sharma, S. D., Mela, A., Merricks, E. M., ... & Canoll, P. (2024). Glioma-Induced Alterations in Excitatory Neurons are Reversed by mTOR Inhibition. bioRxiv.
Bowen, Z., Shilling-Scrivo, K., Losert, W., & Kanold, P. O. (2024). Fractured columnar small-world functional network organization in volumes of L2/3 of mouse auditory cortex. PNAS nexus, 3(2), pgae074.
Znamenskiy, P., Kim, M. H., Muir, D. R., Iacaruso, M. F., Hofer, S. B., & Mrsic-Flogel, T. D. (2024). Functional specificity of recurrent inhibition in visual cortex. Neuron, 112(6), 991-1000.
Ribeiro, T. L., Jendrichovsky, P., Yu, S., Martin, D. A., Kanold, P. O., Chialvo, D. R., & Plenz, D. (2024). Trial-by-trial variability in cortical responses exhibits scaling of spatial correlations predicted from critical dynamics. Cell reports, 43(2).
2023
Roethler, O., Zohar, E., Malina, K. C.-K., Bitan, L., Gabel, H. W., & Spiegel, I. (2023). Single genomic enhancers drive experience-dependent GABAergic plasticity to maintain sensory processing in the adult cortex. Neuron, 111(17), 2693-2708.
Bouchatta, O., Brodzki, M., Manouze, H., Carballo, G. B., Kindström, E., de-Faria, F. M., ... & Szczot, M. (2023). PIEZO2-dependent rapid pain system in humans and mice. bioRxiv, 2023-12.
Lu, T. Y., Hanumaihgari, P., Hsu, E. T., Agarwal, A., Kawaguchi, R., Calabresi, P. A., & Bergles, D. E. (2023). Norepinephrine modulates calcium dynamics in cortical oligodendrocyte precursor cells promoting proliferation during arousal in mice. Nature Neuroscience, 26(10), 1739-1750.
Das, A., Holden, S., Borovicka, J., Icardi, J., O’Niel, A., Chaklai, A., ... & Dana, H. (2023). Large-scale recording of neuronal activity in freely-moving mice at cellular resolution. Nature Communications, 14(1), 6399.
O’Toole, S. M., Oyibo, H. K., & Keller, G. B. (2023). Molecularly targetable cell types in mouse visual cortex have distinguishable prediction error responses. Neuron, 111(18), 2918-2928.
Wool, L., Lak, A., Carandini, M., & Harris, K. (2023). Mouse frontal cortex nonlinearly encodes stimuli, choices, and outcomes. Wellcome Open Research, 8(451), 451.
Sela, M., Poley, M., Mora-Raimundo, P., Kagan, S., Avital, A., Kaduri, M., ... & Schroeder, A. (2023). Brain-targeted liposomes loaded with monoclonal antibodies reduce alpha-synuclein aggregation and improve behavioral symptoms in Parkinson's disease. Advanced Materials, 35(51), 2304654.
Hussain, R., Tithof, J., Wang, W., Cheetham-West, A., Song, W., Peng, W., ... & Nedergaard, M. (2023). Potentiating glymphatic drainage minimizes post-traumatic cerebral oedema. Nature, 623(7989), 992-1000.
Barth-Maron, A., D’Alessandro, I., & Wilson, R. I. (2023). Interactions between specialized gain control mechanisms in olfactory processing. Current Biology, 33(23), 5109-5120.
Chang, H., Esteves, I. M., Neumann, A. R., Mohajerani, M. H., & McNaughton, B. L. (2023). Cortical reactivation of spatial and non-spatial features coordinates with hippocampus to form a memory dialogue. Nature communications, 14(1), 7748.
Ciabatti, E., González-Rueda, A., de Malmazet, D., Lee, H., Morgese, F., & Tripodi, M. (2023). Genomic stability of self-inactivating rabies. eLife, 12, e83459.
Fiore, F., Alhalaseh, K., Dereddi, R. R., Bodaleo Torres, F., Çoban, I., Harb, A., & Agarwal, A. (2023). Norepinephrine regulates calcium signals and fate of oligodendrocyte precursor cells in the mouse cerebral cortex. Nature Communications, 14(1), 8122.
Tsuji, M., Nishizuka, Y. & Emoto, K. (2023). Threat gates visual aversion via theta activity in Tachykinergic neurons. Nature Communications, 14(1), 3987.
Villanueva, C. B., Stephensen, H. J. T., Mokso, R., Benraiss, A., Sporring, J., Goldman, S.A. (2023). Astrocytic engagement of the corticostriatal synaptic cleft is disrupted in a mouse model of Huntington's disease. Proceedings of the National Academy of Sciences, 120(24), e2210719120.
Holstein-Rønsbo, S., Gan, Y., Giannetto, M.J. et al. (2023). Glymphatic influx and clearance are accelerated by neurovascular coupling. Nature Neuroscience, 26, 1042–1053.
Serradj, N., Marino, F., Moreno-López, Y. et al. (2023). Task-specific modulation of corticospinal neuron activity during motor learning in mice. Nature Communications, 14(1), 2708.
Inayat, S., McAllister, B. B., Whishaw, I. Q., & Mohajerani, M. H. (2023). Hippocampal conjunctive and complementary CA1 populations relate sensory events to movement. Iscience, 26(4), 106481.
Untiet, V., Beinlich, F. R., Kusk, P., Kang, N., Ladrón-de-Guevara, A., Song, W., ... & Nedergaard, M. (2023). Astrocytic chloride is brain state dependent and modulates inhibitory neurotransmission in mice. Nature Communications, 14(1), 1871.
Anthoney, N., Tainton-Heap, L., Luong, H., Notaras, E., Zhao, Q., Perry, T., ... & van Swinderen, B. (2023). Experimentally induced active and quiet sleep engage non-overlapping transcriptomes in Drosophila. bioRxiv, 535331.
Boster, K. A., Cai, S., Ladrón-de-Guevara, A., Sun, J., Zheng, X., Du, T., ... & Kelley, D. H. (2023). Artificial intelligence velocimetry reveals in vivo flow rates, pressure gradients, and shear stresses in murine perivascular flows. Proceedings of the National Academy of Sciences, 120(14), e2217744120.
Scarpetta, V., Bodaleo, F., Salio, C., Agarwal, A., Sassoè-Pognetto, M., & Patrizi, A. (2023). Morphological and mitochondrial changes in murine choroid plexus epithelial cells during healthy aging. Fluids and Barriers of the CNS, 20(1), 1-16.
McDermott, K. D., Frechou, M. A., Jordan, J. T., Martin, S. S., & Gonçalves, J. T. (2023). Delayed formation of neural representations of space in aged mice. bioRxiv, 531021.
Matthews, M. D., Cook, E., Naguib, ,N., Wiesner, U. B., & Lewis, K. J. (2023). Intravital imaging of osteocyte integrin dynamics with locally injectable fluorescent nanoparticles. Bone, 174, 116830.
Tort-Colet, N., Resta, F., Montagni, E., Pavone, F., Allegra Mascaro, A. L., & Destexhe, A. (2023). Assessing brain state and anesthesia level with two-photon calcium signals. Scientific Reports, 13(1), 3183.
Esteves, I. M., Chang, H., Neumann, A. R., & McNaughton, B. L. (2023). Consolidation of cellular memory representations in superficial neocortex. Iscience, 26(2), 105970.
Quezada, A., Ward, C., Bader, E. R., Zolotavin, P., Altun, E., Hong, S., ... & Hébert, J. M. (2023). An in vivo platform for rebuilding functional neocortical tissue. Bioengineering, 10(2), 263.
Yang, J. Y., O’Connell, T. F., Hsu, W. M. M., Bauer, M. S., Dylla, K. V., Sharpee, T. O., & Hong, E. J. (2023). Restructuring of olfactory representations in the fly brain around odor relationships in natural sources. bioRxiv, 528627.
Carlton, A. J., Jeng, J. Y., Grandi, F. C., De Faveri, F., Ceriani, F., De Tomasi, L., ... & Marcotti, W. (2023). A critical period of prehearing spontaneous Ca2+ spiking is required for hair-bundle maintenance in inner hair cells. The EMBO Journal, 42(4), e112118.
Hamling, K. R., Zhu, Y., Auer, F., & Schoppik, D. (2023). Tilt In Place Microscopy (TIPM): a simple, low-cost solution to image neural responses to body rotations. Journal of Neuroscience, 43(6), 936-948.
Vestergaard, M., Carta, M., Güney, G., & Poulet, J. F. A. (2023). The cellular coding of temperature in the mammalian cortex. Nature, 614(7949), 725-731.
Contreras-López, R., Alatriste-León, H., Díaz-Hernández, E., Ramírez-Jarquín, J. O., & Tecuapetla, F. (2023). The deep cerebellar nuclei to striatum disynaptic connection contributes to skilled forelimb movement. Cell Reports, 42(1), 112000.
Mabuchi, Y., Cui, X., Xie, L., Kim, H., Jiang, T., & Yapici, N. (2023). GABA-mediated inhibition in visual feedback neurons fine-tunes Drosophila male courtship. bioRxiv, 525544.
Das, A., Margevicius, D., Borovicka, J., Icardi, J., Patel, D., Paquet, M. E., & Dana, H. (2023). Enhanced detection sensitivity of neuronal activity patterns using CaMPARI1 vs. CaMPARI2. Frontiers in Neuroscience, 16, 2291.
Lee, S., Lee, K., Choi, M., & Park, J. (2023). Implantable acousto-optic window for monitoring ultrasound-mediated neuromodulation in vivo. Neurophotonics, 9(3), 032203.
Zhuo, G. Y., Chen, M. C., Lin, T. Y., Lin, S. T., Chen, D. T. L., & Lee, C. W. S. (2023). Opioid-Modulated Receptor Localization and Erk1/2 Phosphorylation in Cells Coexpressing μ-Opioid and Nociceptin Receptors. International Journal of Molecular Sciences, 24(2), 1048.
Diaz-Cuadros, M., Miettinen, T. P., Skinner, O. S., Sheedy, D., Díaz-García, C. M., Gapon, S., ... & Pourquié, O. (2023). Metabolic regulation of species-specific developmental rates. Nature, 613(7944), 550-557.
Evrard, M., Mansuryan, T., Couderc, V., Désévédavy, F., Strutynski, C., Dussauze, M., ... & Smektala, F. (2023). Highly Nonlinear Multimode Tellurite Fibers: From Glass Synthesis to Practical Applications in Multiphoton Imaging. Advanced Photonics Research, 4(1), 2200213.
2022
Graham, R. T., Parrish, R. R., Alberio, L., Johnson, E. L., Owens, L., & Trevelyan, A. J. (2022). Optogenetic stimulation reveals a latent tipping point in cortical networks during ictogenesis. Brain, awac487.
Fisher, Y. E., Marquis, M., D’Alessandro, I., & Wilson, R. I. (2022). Dopamine promotes head direction plasticity during orienting movements. Nature, 612(7939), 316-322.
Wang, X., Delle, C., Asiminas, A., Akther, S., Vittani, M., Brxxxgger, P., ... & Hirase, H. (2022). Liver-secreted fluorescent blood plasma markers enable chronic imaging of the microcirculation. Cell Reports Methods, 2(10), 100302.
Lu, Y., Wei, X., Li, W., Wu, X., Chen, C., Li, G., ... & Gan, W. B. (2022). Large-volume and deep brain imaging in rabbits and monkeys using COMPACT two-photon microscopy. Scientific Reports, 12(1), 17736.
Huang, J. S., Kunkhyen, T., Rangel, A. N., Brechbill, T. R., Gregory, J. D., Winson-Bushby, E. D., ... & Cheetham, C. E. (2022). Immature olfactory sensory neurons provide behaviourally relevant sensory input to the olfactory bulb. Nature Communications, 13(1), 6194.
Govindaraju, I., Muraleedharan, M., Maidin, S., Chakraborty, I., Zhuo, G. Y., Mahato, K. K., & Mazumder, N. (2022). Investigation of the effect of gamma irradiation on the morphological structures of starch using advanced microscopic techniques. In Frontiers in Optics, JTu5A-74.
Hwang, F. J., Roth, R. H., Wu, Y. W., Sun, Y., Kwon, D. K., Liu, Y., & Ding, J. B. (2022). Motor learning selectively strengthens cortical and striatal synapses of motor engram neurons. Neuron, 110(17), 2790-2801.
Yue, Y., Ash, R. T., Boyle, N., Kinter, A., Li, Y., Zeng, C., & Lu, H. (2022). MeCP2 deficiency impairs motor cortical circuit flexibility associated with motor learning. Molecular Brain, 15(1), 1-12.
Liu, Z., Lu, X., Villette, V., Gou, Y., Colbert, K. L., Lai, S., Guan, S., ... & St-Pierre, F. (2022). Sustained deep-tissue voltage recording using a fast indicator evolved for two-photon microscopy. Cell, 185(18), 3408-3425.
Aragon, M. J., Mok, A. T., Shea, J., Wang, M., Kim, H., Barkdull, N., ... & Yapici, N. (2022). Multiphoton imaging of neural structure and activity in Drosophila through the intact cuticle. eLife, 11, e69094.
Matheson, A. M., Lanz, A. J., Medina, A. M., Licata, A. M., Currier, T. A., Syed, M. H., & Nagel, K. I. (2022). A neural circuit for wind-guided olfactory navigation. Nature Communications, 13(1), 4613.
Chornyy, S., Borovicka, J. A., Patel, D., Shin, M. K., Vázquez-Rosa, E., Miller, E., ... & Dana, H. (2022). Longitudinal in vivo monitoring of axonal degeneration after brain injury. Cell Reports Methods, 3(5), 100481.
Sheng, W., Zhao, X., Huang, X., & Yang, Y. (2022). Real-time image processing toolbox for all-optical closed-loop control of neuronal activities. Frontiers in Cellular Neuroscience, 16, 917713.
Tadres, D., Shiozaki, H. M., Tastekin, I., Stern, D. L., & Louis, M. (2022). An essential experimental control for functional connectivity mapping with optogenetics. bioRxiv, 493610.
Koveal, D., Rosen, P. C., Meyer, D. J., Díaz-García, C. M., Wang, Y., Cai, L. H., ... & Yellen, G. (2022). A high-throughput multiparameter screen for accelerated development and optimization of soluble genetically encoded fluorescent biosensors. Nature Communications, 13(1), 2919.
Fukuda, M., Matsumura, T., Suda, T., & Hirase, H. (2022). Depth-targeted intracortical microstroke by two-photon photothrombosis in rodent brain. Neurophotonics, 9(2), 021910-021910.
Jackson, J. G., Krizman, E., Takano, H., Lee, M., Choi, G. H., Putt, M. E., & Robinson, M. B. (2022). Activation of Glutamate Transport Increases Arteriole Diameter in vivo: Implications for Neurovascular Coupling. Frontiers in Cellular Neuroscience, 16, 831061.
Du, T., Mestre, H., Kress, B. T., Liu, G., Sweeney, A. M., Samson, A. J., ... & Nedergaard, M. (2022). Cerebrospinal fluid is a significant fluid source for anoxic cerebral oedema. Brain, 145(2), 787-797.
Govindaraju, I., Zhuo, G. Y., Chakraborty, I., Melanthota, S. K., Mal, S. S., Sarmah, B., ... & Mazumder, N. (2022). Investigation of structural and physico-chemical properties of rice starch with varied amylose content: A combined microscopy, spectroscopy, and thermal study. Food Hydrocolloids, 122, 107093.
Choe, K., Hontani, Y., Wang, T., Hebert, E., Ouzounov, D. G., Lai, K., ... & Xu, C. (2022). Intravital three-photon microscopy allows visualization over the entire depth of mouse lymph nodes. Nature Immunology, 23(2), 330-340.
Spivak, Y. M., & Lemeshko, P. S. (2022). Multiphoton microscopy of mesoporous silicon. In Journal of Physics: Conference Series. 2227(1), 012013.
2021
Maset, A., Albanesi, M., di Soccio, A., Canova, M., Dal Maschio, M., & Lodovichi, C. (2021). Aberrant Patterns of Sensory-Evoked Activity in the Olfactory Bulb of LRRK2 Knockout Mice. Cells, 10(11), 3212.
Schmidt, E. R., Zhao, H. T., Park, J. M., Dipoppa, M., Monsalve-Mercado, M. M., Dahan, J. B., ... & Polleux, F. (2021). A human-specific modifier of cortical connectivity and circuit function. Nature, 599(7886), 640-644.
Hermans, L., Kaynak, M., Braun, J., Lobato Ríos, V., Chen, C. L., Günel, S., ... & Ramdya, P. (2021). Long-term imaging of the ventral nerve cord in behaving adult Drosophila. bioRxiv, 463778.
Carlsen, E. M. M., Falk, S., Skupio, U., Robin, L., Pagano Zottola, A. C., Marsicano, G., & Perrier, J. F. (2021). Spinal astroglial cannabinoid receptors control pathological tremor. Nature Neuroscience, 24(5), 658-666.
Moussa, N. O., Mansuryan, T., Hage, C. H., Fabert, M., Krupa, K., Tonello, A., ... & Couderc, V. (2021). Spatiotemporal beam self-cleaning for high-resolution nonlinear fluorescence imaging with multimode fiber. Scientific Reports, 11(1), 18240.
Eleftheriou, C. G., Corona, C., Khattak, S., Alam, N. M., Ivanova, E., Bianchimano, P., ... & Sagdullaev, B. T. (2021). Retinoschisin deficiency induces persistent aberrant waves of activity affecting neuroglial signaling in the retina. Journal of Neuroscience, 42(36), 6983-7000.
Han, J., Kim, S., Choi, P., Lee, S., Jo, Y., Kim, E., & Choi, M. (2021). Robust functional imaging of taste sensation with a Bessel beam. Biomedical Optics Express, 12(9), 5855-5864.
Favuzzi, E., Huang, S., Saldi, G. A., Binan, L., Ibrahim, L. A., Fernández-Otero, M., ... & Fishell, G. (2021). GABA-receptive microglia selectively sculpt developing inhibitory circuits. Cell, 184(15), 4048-4063.
Chornyy, S., Das, A., Borovicka, J. A., Patel, D., Chan, H. H., Hermann, J. K., ... & Dana, H. (2021). Cellular-resolution monitoring of ischemic stroke pathologies in the rat cortex. Biomedical Optics Express, 12(8), 4901-4919.
Malina, K. C. K., Tsivourakis, E., Kushinsky, D., Apelblat, D., Shtiglitz, S., Zohar, E., ... & Spiegel, I. (2021). NDNF interneurons in layer 1 gain-modulate whole cortical columns according to an animal’s behavioral state. Neuron, 109(13), 2150-2164.
Bruzzone, M., Chiarello, E., Albanesi, M., Miletto Petrazzini, M. E., Megighian, A., Lodovichi, C., & Dal Maschio, M. (2021). Whole brain functional recordings at cellular resolution in zebrafish larvae with 3D scanning multiphoton microscopy. Scientific Reports, 11(1), 11048.
Mridha, Z., de Gee, J. W., Shi, Y., Alkashgari, R., Williams, J., Suminski, A., ... & McGinley, M. J. (2021). Graded recruitment of pupil-linked neuromodulation by parametric stimulation of the vagus nerve. Nature Communications, 12(1), 1539.
Ivanova, E., Corona, C., Eleftheriou, C. G., Bianchimano, P., & Sagdullaev, B. T. (2021). Retina-specific targeting of pericytes reveals structural diversity and enables control of capillary blood flow. Journal of Comparative Neurology, 529(6), 1121-1134.
Hung, C. W., Mazumder, N., Lin, D. J., Chen, W. L., Lin, S. T., Chan, M. C., & Zhuo, G. Y. (2021). Label-free characterization of collagen crosslinking in bone-engineered materials using nonlinear optical microscopy. Microscopy and Microanalysis, 27(3), 587-597.
Hontani, Y., Xia, F., & Xu, C. (2021). Multicolor three-photon fluorescence imaging with single-wavelength excitation deep in mouse brain. Science Advances, 7(12), eabf3531.
Resnik, J., & Polley, D. B. (2021). Cochlear neural degeneration disrupts hearing in background noise by increasing auditory cortex internal noise. Neuron, 109(6), 984-996.
Tainton-Heap, L. A., Kirszenblat, L. C., Notaras, E. T., Grabowska, M. J., Jeans, R., Feng, K., ... & van Swinderen, B. (2021). A paradoxical kind of sleep in Drosophila melanogaster. Current Biology, 31(3), 578-590.
Hortholary, T., Carrion, C., Chouzenoux, E., Pesquet, J. C., & Lefort, C. (2021). Multiplex-multiphoton microscopy and computational strategy for biomedical imaging. Microscopy Research and Technique, 84(7), 1553-1562.
Clayton, K. K., Williamson, R. S., Hancock, K. E., Tasaka, G. I., Mizrahi, A., Hackett, T. A., & Polley, D. B. (2021). Auditory corticothalamic neurons are recruited by motor preparatory inputs. Current Biology, 31(2), 310-321.
Esteves, I. M., Chang, H., Neumann, A. R., Sun, J., Mohajerani, M. H., & McNaughton, B. L. (2021). Spatial information encoding across multiple neocortical regions depends on an intact hippocampus. Journal of Neuroscience, 41(2), 307-319.
Schumacher, J. W., McCann, M. K., Maximov, K. J., & Fitzpatrick, D. (2021). Selective enhancement of neural coding in V1 underlies fine-discrimination learning in tree shrew. Current Biology, 32(15), 3245-3260.
2020
Keller, A. J., Dipoppa, M., Roth, M. M., Caudill, M. S., Ingrosso, A., Miller, K. D., & Scanziani, M. (2020). A disinhibitory circuit for contextual modulation in primary visual cortex. Neuron, 108(6), 1181-1193.
Lu, J., Behbahani, A. H., Hamburg, L., Westeinde, E. A., Dawson, P. M., Lyu, C., ... & Wilson, R. I. (2020). Transforming representations of movement from body-to world-centric space. Nature, 601(7891), 98-104.
Scholl, B., Thomas, C. I., Ryan, M. A., Kamasawa, N., & Fitzpatrick, D. (2020). Cortical response selectivity derives from strength in numbers of synapses. Nature, 590(7844), 111-114.
Robinson, N. T., Descamps, L. A., Russell, L. E., Buchholz, M. O., Bicknell, B. A., Antonov, G. K., ... & Häusser, M. (2020). Targeted activation of hippocampal place cells drives memory-guided spatial behavior. Cell, 183(6), 1586-1599.
Fan, J. L., Rivera, J. A., Sun, W., Peterson, J., Haeberle, H., Rubin, S., & Ji, N. (2020). High-speed volumetric two-photon fluorescence imaging of neurovascular dynamics. Nature Communications, 11(1), 6020.
Okubo, T. S., Patella, P., D’Alessandro, I., & Wilson, R. I. (2020). A neural network for wind-guided compass navigation. Neuron, 107(5), 924-940.
Mazzarda, F., D'Elia, A., Massari, R., De Ninno, A., Bertani, F. R., Businaro, L., ... & Mammano, F. (2020). Organ-on-chip model shows that ATP release through connexin hemichannels drives spontaneous Ca 2+ signaling in non-sensory cells of the greater epithelial ridge in the developing cochlea. Lab on a Chip, 20(16), 3011-3023.
Bowen, Z., Winkowski, D. E., & Kanold, P. O. (2020). Functional organization of mouse primary auditory cortex in adult C57BL/6 and F1 (CBAxC57) mice. Scientific Reports, 10(1), 10905.
Feng, K., Sen, R., Minegishi, R., Dübbert, M., Bockemühl, T., Büschges, A., & Dickson, B. J. (2020). Distributed control of motor circuits for backward walking in Drosophila. Nature Communications, 11(1), 6166.
Keller, A. J., Roth, M. M., & Scanziani, M. (2020). Feedback generates a second receptive field in neurons of the visual cortex. Nature, 582(7813), 545-549.
Eschbach, C., Fushiki, A., Winding, M., Afonso, B., Andrade, I. V., Cocanougher, B. T., ... & Zlatic, M. (2020). Circuits for integrating learnt and innate valences in the fly brain. bioRxiv, 058339.
Montgomery, M. K., Kim, S. H., Dovas, A., Zhao, H. T., Goldberg, A. R., Xu, W., ... & Hillman, E. M. (2020). Glioma-induced alterations in neuronal activity and neurovascular coupling during disease progression. Cell Reports, 31(2), 107500.
Shiozaki, H. M., Ohta, K., & Kazama, H. (2020). A multi-regional network encoding heading and steering maneuvers in Drosophila. Neuron, 106(1), 126-141.
Mestre, H., Du, T., Sweeney, A. M., Liu, G., Samson, A. J., Peng, W., ... & Nedergaard, M. (2020). Cerebrospinal fluid influx drives acute ischemic tissue swelling. Science, 367(6483), eaax7171.
Jeng, J. Y., Ceriani, F., Hendry, A., Johnson, S. L., Yen, P., Simmons, D. D., ... & Marcotti, W. (2020). Hair cell maturation is differentially regulated along the tonotopic axis of the mammalian cochlea. The Journal of Physiology, 598(1), 151-170.
Kovacs-Oller, T., Ivanova, E., Bianchimano, P., & Sagdullaev, B. T. (2020). The pericyte connectome: spatial precision of neurovascular coupling is driven by selective connectivity maps of pericytes and endothelial cells and is disrupted in diabetes. Cell Discovery, 6(1), 39.
Oe, Y., Wang, X., Patriarchi, T., Konno, A., Ozawa, K., Yahagi, K., ... & Hirase, H. (2020). Distinct temporal integration of noradrenaline signaling by astrocytic second messengers during vigilance. Nature Communications, 11(1), 471.
2019
Romero, S., Hight, A. E., Clayton, K. K., Resnik, J., Williamson, R. S., Hancock, K. E., & Polley, D. B. (2019). Cellular and widefield imaging of sound frequency organization in primary and higher order fields of the mouse auditory cortex. Cerebral Cortex, 30(3), 1603-1622.
Nardin, C., Peres, C., Mazzarda, F., Ziraldo, G., Salvatore, A. M., & Mammano, F. (2019). Photosensitizer activation drives apoptosis by interorganellar Ca2+ transfer and superoxide production in bystander cancer cells. Cells, 8(10), 1175.
Scholl, B., Wilson, D. E., Jaepel, J., & Fitzpatrick, D. (2019). Functional logic of layer 2/3 inhibitory connectivity in the ferret visual cortex. Neuron, 104(3), 451-457.
Ceriani, F., Johnson, S. L., Sedlacek, M., Hendry, A., Kachar, B., Marcotti, W., & Mammano, F. (2019). Dynamic coupling of cochlear inner hair cell intrinsic Ca2+ action potentials to Ca2+ signaling of non-sensory cells. bioRxiv, 731851.
Brawek, B and Garaschuk O. (2019). Single-cell electroporation for measuring in vivo calcium dynamics in microglia. Microglia Methods and Protocols, 231-241.
Brawek, B., Olmedillas del Moral, M., & Garaschuk, O. (2019). In vivo visualization of microglia using tomato lectin. Microglia Methods and Protocols, 165-175.
Liang, Y. & Garaschuk O. (2019). Labeling microglia with genetically encoded calcium indicators. Microglia Methods and Protocols, 243–265.
Royzen, F., Williams, S., Fernandez, F. R., & White, J. A. (2019). Balanced synaptic currents underlie low-frequency oscillations in the subiculum. Hippocampus, 29(12), 1178-1189.
Rathore, A. P. S., Mantri, C. K., Aman, S. A. B., Syenina, A., Ooi, J., Jagaraj, C. J., ... & St. John, A. L. (2019). Dengue virus-elicited tryptase induces endothelial permeability and shock. Journal of Clinical Investiagtion, 129(10), 4180-4193.
Stringer, C., Pachitariu, M., Steinmetz, N., Carandini, M., & Harris, K. D. (2019). High-dimensional geometry of population responses in visual cortex. Nature, 571(7765), 361–365.
Ziraldo, G., Buratto, D., Kuang, Y., Xu, L., Carrer, A., Nardin, C., ... & Mammano, F. (2019). A human-derived monoclonal antibody targeting extracellular connexin domain selectively modulates hemichannel function. Frontiers in Physiology, 10, 392.
Díaz-García, C. M., Lahmann, C., Martínez-François, J. R., Li, B., Koveal, D., Nathwani N., ... & Yellen, G. (2019). Quantitative in vivo imaging of neuronal glucose concentrations with a genetically encoded fluorescence lifetime sensor. Journal of Neuroscience Research, 97(8), 946–960.
Philip, V., Newton, D., Oh, H., Collins, S., Bercik, P., & Sibille, E. (2019). The Effect of Gut Microbiota on Glutamatergic/GABAergic Gene Expression in Adult Mice. Biological Psychiatry, 85, S127–S128.
Bowen, Z., Winkowski, D. E., Seshadri, S., Plenz, D., & Kanold, P. O. (2019). Neuronal avalanches in input and associative layers of auditory cortex. Frontiers in Systems Neuroscience, 13, 45.
Burgold, J., Schulz-Trieglaff, E. K., Voelkl, K., Gutiérrez-Ángel, S., Bader, J. M., Hosp, F., ... & Dudanova, I. (2019). Cortical circuit alterations precede motor impairments in Huntington’s disease mice. Scientific Reports, 9(1), 6634.
Matovic, S., Ichiyama, A., Igarashi, H., Salter, E. W., Wang, X. F., Henry, M., ... & Inoue, W. (2019). Stress-induced neuronal hypertrophy decreases the intrinsic excitability in stress habituation. bioRxiv, 593665.
Weissenberger, Y., King, A. J., & Dahmen, J. C. (2019). Decoding mouse behavior to explain single-trial decisions and their relationship with neural activity. bioRxiv, 567479.
Ceriani, F., Hendry, A., Jeng, J. Y., Johnson, S. L., Stephani, F., Olt, J., ... & Marcotti, W. (2019). Coordinated calcium signalling in cochlear sensory and non-sensory cells refines afferent innervation of outer hair cells. The EMBO Journal, 38(9), e99839.
Lee, K. S., Vandemark, K., Mezey, D., Shultz, N., & Fitzpatrick, D. (2019). Functional synaptic architecture of callosal inputs in mouse primary visual cortex. Neuron, 101(3), 421-428.
2018
Marvin, J. S., Scholl, B., Wilson, D. E., Podgorski, K., Kazemipour, A., Müller, J. A., ... & Looger, L. L. (2018). Stability, affinity, and chromatic variants of the glutamate sensor iGluSnFR. Nature Methods, 15(11), 936–939.
Moeyaert, B., Holt, G., Madangopal, R., Perez-Alvarez, A., Fearey, B. C., Trojanowski, N. F., ... & Schreiter, E. R. (2018). Improved methods for marking active neuron populations. Nature Communications, 9(1), 440.
Chen, C. L., Hermans, L., Viswanathan, M. C., Fortun, D., Aymanns, F., Unser, M., ... & Ramdya, P. (2018). Imaging neural activity in the ventral nerve cord of behaving adult Drosophila. Nature Communications, 9(1), 4390.
Corns, L. F., Johnson, S. L., Roberts, T., Ranatunga, K. M., Hendry, A., Ceriani, F., ... & Marcotti, W. (2018). Mechanotransduction is required for establishing and maintaining mature inner hair cells and regulating efferent innervation. Nature Communications, 9(1), 4015.
Saleem, A. B., Diamanti, E. M., Fournier, J., Harris, K. D., & Carandini, M. (2018). Coherent encoding of subjective spatial position in visual cortex and hippocampus. Nature, 562(7725), 124-127.
Dipoppa, M., Ranson, A., Krumin, M., Pachitariu, M., Carandini, M., & Harris, K. D. (2018). Vision and locomotion shape the interactions between neuron types in mouse visual cortex. Neuron, 98(3), 602-615.
Gillet, S. N., Kato, H. K., Justen, M. A., Lai, M., & Isaacson, J. S. (2018). Fear learning regulates cortical sensory representations by suppressing habituation. Frontiers in Neural Circuits, 11, 112.
2017
Scholl, B., Wilson, D. E., & Fitzpatrick, D. (2017). Local order within global disorder: synaptic architecture of visual space. Neuron, 96(5), 1127-1138.
Klapoetke, N. C., Nern, A., Peek, M. Y., Rogers, E. M., Breads, P., Rubin, G. M., ... & Card, G. M. (2017). Ultra-selective looming detection from radial motion opponency. Nature Neuroscience, 551(7679), 237-241.
Kato, H. K., Asinof, S. K., & Isaacson, J. S. (2017). Network-level control of frequency tuning in auditory cortex. Neuron, 95(2), 412-423.
Lu, R., Sun, W., Liang, Y., Kerlin, A., Bierfeld, J., Seelig, J.D., W... & Ji, N. (2017). Video-rate volumetric functional imaging of the brain at synaptic resolution. Nature Neuroscience, 20(4), 620-628.
2016
Mongeon, R., Venkatachalam, V., & Yellen, G. (2016). Cytosolic NADH-NAD+ redox visualized in brain slices by two-photon fluorescence lifetime biosensor imaging.. Antioxid Redox Signal, 25(10), 553-563.
Pachitariu, M., Stringer, C., Schröder, S., Dipoppa, M., Rossi, L. F., Carandini, M., & Harris, K. D. (2016). Suite2p: beyond 10,000 neurons with standard two-photon microscopy. bioRxiv, 061507.
Rose, T., Jaepel, J., Hübener, M., & Bonhoeffer, T. (2016). Cell-specific restoration of stimulus preference after monocular deprivation in the visual cortex. Science, 352(6291), 1319–1322.
Strobl, M. J., Freeman, D., Patel, J., Poulsen, R., Wendler, C. C., Rivkees, S. A., & Coleman, J. E. (2016). Opposing effects of maternal hypo-and hyperthyroidism on the stability of thalamocortical synapses in the visual cortex of adult offspring.. Cerebral Cortex, 27(5), 3015-3027.
Lee, K. S., Huang, X., & Fitzpatrick, D. (2016). Topology of ON and OFF inputs in visual cortex enables an invariant columnar architecture. Nature, 533(7601), 90-94.
Monai, H., Ohkura, M., Tanaka, M., Oe, Y., Konno, A., Hirai, H., ... & Hirase, H. (2016). Calcium imaginq reveals glial involvement in transcranial direct current stimulation-induced plasticity in mouse brain. Nature Communications, 7(1), 11100.
Ganmor, E., Krumin, M., Rossi, L. F., Carandini, M., & Simoncelli, E. P. (2016). Direct estimation of firing rates from calcium imaging data. arXiv, 1601.00364.
2015
Roth, M. M., Dahmen, J.C., Muir, D. R., Imhof, F., Martini, F. J., & Hofer, S. B. (2015). Thalamic nuclei convey diverse contextual information to layer 1 of visual cortex. Nature Neuroscience, 19(2), 299-307.
Barnstedt, O., Keating, P., Weissenberger, Y., King, A. J., & Dahmen, J. C. (2015). Functional microarchitecture of the mouse dorsal inferior colliculus revealed through in vivo two-photon calcium imaging.. Journal of Neuroscience, 35(31), 10927-10939.
Chen, S. X., Kim, A. N., Peters, A. J., & Komiyama, T. (2015). Subtype-specific plasticity of inhibitory circuits in motor cortex during motor learning. Nature Neuroscience, 18(8), 1109-1115.
Jia, Y., Zhang, S., Miao, L., Wang, J., Jin, Z., Gu, B., ... & Li, Z. (2015). Activation of platelet protease-activated receptor-1 induces epithelial-mesenchymal transition and chemotaxis of colon cancer cell line SW620. Oncology Reports, 33(6), 2681-2688.
Lu, W., Tang, Y., Zhang, Z., Zhang, X., Yao, Y., Fu, C., ... & Ma, G. (2015). Inhibiting the mobilization of Ly6Chigh monocytes after acute myocardial infarction enhances the efficiency of mesenchymal stromal cell transplantation and curbs myocardial remodeling.. American Journal of Translational Research, 7(3), 587-597.
Boyd, A. M., Kato, H. K., Komiyama, T., & Isaacson, J. S. (2015). Broadcasting of cortical activity to the olfactory bulb. Cell Reports, 10(7), 1032-1039.
Cossell, L., Iacaruso, M. F., Muir, D. R., Houlton, R., Sader, E. N., Ko, H., H... & Mrsic-Flogel, T. D. (2015). Functional organization of excitatory synaptic strength in primary visual cortex. Nature, 518(7539), 399-403.
2014
Partridge, J. G., Lewin, A. E., Yasko, J. R., & Vicini, S. (2014). Contrasting actions of group I metabotropic glutamate receptors in distinct mouse striatal neurones. Journal of Physiology, 592(13), 2721-2733.
Peters, A. J., Chen, S, X., Komiyama, T. (2014). Emergence of reproducible spatiotemporal activity during motor learning. Nature, 510(7504), 263-267.
Ehmke, T., Nitzsche, T. H., Knebl, A., & Heisterkamp, A. (2014). Molecular orientation sensitive second harmonic microscopy by radially and azimuthally polarized light. Biomedical Optics Express, 5(7), 2231-46.
Liu, J., Wu, N., Ma, L., Liu, M., Liu, G., Zhang, Y., & Lin, X. (2014). Oleanolic acid suppresses aerobic glycolysis in cancer cells by switching pyruvate kinase type M isoforms. PLoS One, 9(3), e91606.
Palmer, L. M., Shai, A. S., Reeve, J.E., Anderson, H. L., Paulsen, O., & Larkum, M. E. (2014). NMDA spikes enhance action potential generation during sensory input. Nature Neuroscience, 17(3), 383-390.
Cai, F., Yu, J., Qian, J., Wang, Y., Chen, Z., Huang, J., ... & He, S. (2014). Use of tunable second-harmonic signal from KNbO3 nanoneedles to find optimal wavelength for deep-tissue imaging. Laser & Photonics Reviews, 8(6), 865-874.
2013
Kato, H. K., Gillet, S. N., Peters, A. J., Isaacson, J. S., & Komiyama, T. (2013). Parvalbumin-expressing interneurons linearly control olfactory bulb output. Neuron, 80(5), 1218-1231.
Takata, N., Nagai, T., Ozawa, K., Oe, Y., Mikoshiba, K., & Hirase, H. (2013). Cerebral blood flow modulation by Basal forebrain or whisker stimulation can occur independently of large cytosolic Ca2+ signaling in astrocytes. PLoS One, 8(6), e66525.
![]()
ThorImage®LSのソースコードは、Bergamo®、Cerna® 対応共焦点顕微鏡、Veneto®または共焦点顕微鏡をお持ちのお客様にご提供可能です。当社にメールにてご連絡ください。
ThorImage®LSソフトウェア
ThorImageLSは、当社の顕微鏡ならびに補助的な外付けハードウェアを制御するオープンソースの画像取得プログラムです。切片の多光子Zスタックからin vivoの同時光刺激やイメージングまで、ThorImageLSはそれぞれのニーズに合わせて組み込まれたモジュール式のワークスペースをご提供しております。そのワークフロー指向のインターフェイスは、単一画像、Zスタック、タイムシリーズ、そしてストリーミング画像の取得、可視化ならびに解析をサポートします。ThorImageLSのデータ取得ならびに解析の様子がVideo 121Aでご覧いただけます。
ThorImageLSは当社の顕微鏡をお買い求めいただくと付属しています。またオープンソースのため、ソフトウェア機能や性能の完全カスタマイズが可能です。ThorImageLSには当社のカスタマーサポートならびに定期的なソフトウェア更新サービスが付帯しており、常にイメージングの需要に合うよう心がけております。
詳細については製品紹介ページをご覧ください。
高機能ソフトウェア
- カスタマイズ可能な複数の表示欄を持つワークスペース
- ハードウェア入力とタイミング同期した画像取得
- ライブ画像の補正と関心領域の解析
- ガルバノ-ガルバノならびにガルバノ-レゾナント走査の領域・形状の個別設定
- タイリングによる高分解能広域イメージング
- 高速組織深部スキャンに適した1次、2次Z軸の個別制御
- スクリプトを使用した自動画像キャプチャ
- ImageJ Macrosに対応
- ワークステーション共有時にもマルチユーザの設定を保存
- 検出チャンネル毎に異なるカラー表示で簡単なビジュアル解析
実験でのシームレスな組み込み
- 空間変調モジュールを使用した同時多点光刺激とイメージング
- PFM450Eまたはサードパーティの対物レンズスキャナを使用した高速Zスタック取得
- 電気生理学のための信号制御
- Coherent社のChameleonレーザを使用した波長切り替え
- ポッケルスセルによる関心領域のマスキング
- 深度に応じたパワーランプ制御でダメージを最小に抑制、Signal to Noise 比を最大化
新機能: Version 4.3(クリックしてご覧ください。)
お持ちの顕微鏡に対応する最新のThorImageLSについては当社までお問い合わせください。ThorImageLS 4.xは、バージョン3. x 、2.xならびに1.xに新規機能を大幅に追加しており、旧モデルの顕微鏡に対応できない場合があります。当社では旧モデルをお持ちのお客様のために旧バージョンのソフトウェアのサポートを継続しております。旧バージョンの機能についてはこちらをご覧ください。
- Added Support for the Toptica iChrome CLE-50 Laser
- Added Support for 3P Imaging
- Added Support for the CS126MU, CS165MU, and CC505MU Monochrome Cameras
- Added Support for a Mini-Circuits® Switch Box
- Added Support for PMT3100R (Included in Some Bergamo II Multiphoton Imaging Systems)
- Added Configurable Channel View Layout (Horizontal and Vertical)
- Added Improved Scan Path Realignment and Added Ability to Save Multiple Reference Images for Multiple Targets
- Allows for Simplified Relocation to Same Target Day to Day
- Added New Features for SLM Operation
- Added 3D Mode Toggle
- Added Z Offset
- Added Ability to Export Patterns
- Added Set Zero% and Delete All Buttons
- Added Pattern Center to Display as "0" Point
- Added SLM Control Panel Advanced Mode Enhancement
- Added New Optional Delay Between Epochs
- Added SLM Control Panel Import/Export Enhancement
- Ability to Export Table of SLM Patterns
- Added New SLM Settings
- Added to ThorSLMSettings.xml and Application Settings
- Added Ability to Offset the Z Position of Pattern Points in New 3D Mode
- Enhanced ORCA Fusion Features
- Updated Exposure Calculation for Master Pulse Mode
- Added Option to Enable Water Cooling Control
- Added Improved Performance When Switching Modalities
- Enhanced Two-Way Scanning
- Two-Way Scanning is Now Allowed Up to a Pixel Density of 4096 x 4096 When Only One Channel is Selected
- Only Selectable by Dragging the Slider Bar to the Desired Pixel Density in One-Way Before Switching to Two-Way
- Added Camera Frame Rate Control
- Added UI Control of Two Blue Mini-Circuits® Switch Boxes
- Added 3D SLM
- Added Continuous Preview and Enhanced Orthogonal Views for Z-Stacks
- Added Improved Laser Safety Control for Digital Switches When Switches Are Configured for Laser Switching
- Added Control for Stimulus Shutter Operation When Using Stimulus Capture Mode
- Added Camera Frame Rate Control
- Added a New Section to Control the Frame Rate for CMOS Cameras with this Functionality
- Added Image-Based Autofocus
- Added Ability to Find the Optimal Focus Point of the Sample Based on Image Contrast
- Added Automatic Version Update Checker
- When Connected to Internet a Version Update Check Will Occur as the Splash Screen Loads When ThorImageLS Is Started Up
- Added Signal Generator Analog Mode
- Allows Custom Control of Analog Modulation
- Added New Continuous Button for Repeated Preview
- Allows for Fast Location Sample in XZ and YZ Line Scan Mode
- Added Enhanced IPC Communication
- Added IPC Command to Load an Experiment or a Template
- Added IPC Commands to Move X, Y, Z, and Secondary Z Stages
- Added IPC Commands that Get Sent from ThorImageLS Every Time a File Is Saved During T Series Experiments
Video 121A ThorImage®LSの特長
| Supported Imaging Platforms | ||
|---|---|---|
 Veneto® Inverted Microscopes |  Bergamo® II Multiphoton Microscopes |  Confocal Imaging Systems |
| Laser Scanning | |||
|---|---|---|---|
| Scan Path Wavelength Range | 450 - 1100 nm, 680 - 1300 nm, or 800 - 1800 nm | ||
| Scan Paths | Resonant-Galvo-Galvo Scanner, Galvo-Resonant Scanners, Galvo-Galvo Scanners, or Spatial Light Modulator; Single or Dual Scan Paths | ||
| Scan Speed | 8 kHz Resonant-Galvo-Galvo or Galvo-Resonant | 2 fps at 4096 x 4096 Pixels 30 fps at 512 x 512 Pixels 400 fps at 512 x 32 Pixels | |
| 12 kHz Resonant-Galvo-Galvo or Galvo-Resonant | 4.4 fps at 2048 x 2048 Pixels 45 fps at 512 x 512 Pixels 600 fps at 512 x 32 Pixels | ||
| Galvo-Galvo | 3 fps at 512 x 512 Pixels 48 fps at 512 x 32 Pixels 70 fps at 32 x 32 Pixels Pixel Dwell Time: 0.4 to 20 µs | ||
| Galvo-Galvo Scan Modes | Imaging: Line, Polyline, Square, or Rectangle Non-Imaging: Circle, Ellipse, Polygon, or Point | ||
| Field of View | FN40 Equivalent to 40 mm Diagonal Square (Max) at the Intermediate Image Plane >2.8 mm x 2.8 mm at the Sample plane with a 10X Objective FN20 Equivalent to 20 mm Diagonal Square (Max) at the Intermediate Image Plane >1.4 mm x 1.4 mm at the Sample plane with a 10X Objective | ||
| Scan Zoom | 1X to 16X (Continuously Variable) | ||
| Scan Resolution | Up to 2048 x 2048 Pixels (Bi-Directional) [Up to 1168 x 1168 Pixels for 12 kHz Scanners] Up to 4096 x 4096 Pixels (Unidirectional) [Up to 2336 x 2336 Pixels for 12 kHz Scanners] | ||
| Compatible Objective Threadings | M34 x 1.0, M32 x 0.75, M25 x 0.75, and RMS | ||
| Spatial Light Modulator (SLM) | ||||
|---|---|---|---|---|
| Stimulation Area (with a 16X Objective) | 600 µm x 600 µm (X and Y) ±150 µm (Z) | |||
| Resolution | 1024 pixels x 1024 pixels | |||
| Maximum Pattern Refresh Rate | 4 ms | |||
| Multiphoton Signal Detection | ||
|---|---|---|
| Epi Detection | Up to Four Ultrasensitive GaAsP PMTs, Cooled or Non-Cooled | |
| Forward-Direction Detection | Two Ultrasensitive GaAsP PMTs | |
| Maximum of Four PMTs Controlled by the Software at a Given Time | ||
| Collection Optics | 8°, 10°, or 14° Collection Angle (Angles Quoted When Using an Objective with a 20 mm Entrance Pupil) Easy-to-Exchange Emission Filters and Dichroic Mirrors | |
| Confocal Imaging | ||
|---|---|---|
| Motorized Pinhole Wheel with 16 Round Pinholes from Ø25 µm to Ø2 mm Two to Four Laser Lines (488 nm Standard; Other Options Range from 405 nm to 660 nm) Standard Multialkali or High-Sensitivity GaAsP PMTs Easy-to-Exchange Emission Filters and Dichroic Mirrors |
| Widefield Viewing | ||
|---|---|---|
| Manual or Motorized Switching Between Scanning and Widefield Modes Illumination Provided via LED or Liquid Light Guide C-Mount Threads for Scientific Cameras |
| Transmitted Light Imaging | ||
|---|---|---|
| Differential Interference Contrast (DIC) or Dodt Gradient Contrast Widefield or Laser Scanned Illumination Provided by Visible and/or NIR LEDs Compatible with Air or Oil Immersion Condensers |
| Three-Photon Imaging | ||
|---|---|---|
| Scan Optics for 800 - 1800 nm Range Achieve Reduced Background Scatter for Greater Sensitivity in Deep-Tissue Imaging |
| Volume Imaging Using Bessel Beams | ||
|---|---|---|
| 3D Volumetric Functional Imaging at Video Frame Rates Enhanced Temporal Resolution for Studying Internal Systems at Cellular Lateral Resolution In Vivo |
| Translation | ||||
|---|---|---|---|---|
| Microscope Body Rotation (Rotating Bodies Only) | 0° to 90° or -45° to +45° Around Objective Focus 0.1° Encoder Resolution | |||
| Coarse Elevator Base Z (Rotating Bodies Only) | 5" (127 mm) Total Travel; 1 µm Encoder Resolution | |||
| Fine Microscope Body X and Y | 2" (50.8 mm) Total Travel; 0.5 µm Encoder Resolution | |||
| Fine Microscope Arm Z | 1" (25.4 mm) Total Travel; 0.1 µm Encoder Resolution | |||
| Fine Z Focus | ||||
|---|---|---|---|---|
| Piezo Objective Scanner | Open Loop: 600 µm ± 10% Travel Range; 1 nm Resolution Closed Loop: 450 µm Travel Range; 3 nm Resolution | |||
| Vibrationless Remote Focus | 16 Discrete Steps ~400 µm Travel Range with a 10X Objective ~160 µm Travel Range with a 16X Objective | |||
当社では、用途ごとのさまざまなご要望にお応えできるように、お客様のニーズに合わせたご提案を心掛けています。ご意見・ご要望、またご質問などございましたら当社までお気軽にご連絡ください。

Click to Enlarge
Figure 39A デモルーム例(中国オフィス)
デモルーム・オンラインデモのご案内
ソーラボの技術者は、世界9カ所のオフィスをベースにしており、お客様の実験用途に適したイメージングシステムをお選びいただくためのお手伝いをいたします。生物学のあらゆる課題解決に向けて研究を行うお客様のために、ニーズに合致し、かつ使いやすく、高い信頼性と対応力のあるシステムを提供いたします。
当社では、実際に当社顕微鏡システムなどを無償でお試しいただけるデモルームをご用意しています。オンラインデモも承ります。 デモルームやオンラインデモのご予約、お問い合わせは当社までご連絡ください。
カスタマーサポート(海外)
(クリックすると詳細がご覧いただけます)
Newton, New Jersey, USA
Thorlabs Headquarters
43 Sparta Avenue
Newton, NJ 07860
Customer Support
- Phone: (973) 300-3000
- E-mail: techsupport@thorlabs.com
Ely, United Kingdom
Thorlabs Ltd.
1 Saint Thomas Place, Ely
Ely CB7 4EX
Customer Support
- Phone: +44 (0)1353-654440
- E-mail: techsupport.uk@thorlabs.com
Bergkirchen, Germany
Thorlabs GmbH
Münchner Weg 1
85232 Bergkirchen
Customer Support
- Phone: +49 (0) 8131-5956-0
- E-mail: europe@thorlabs.com
Maisons-Laffitte, France
Thorlabs SAS
109, rue des Cotes
Maisons-Laffitte 78600
Customer Support
- Phone: +33 (0)970 440 844
- E-mail: techsupport.fr@thorlabs.com
São Carlos, SP, Brazil
Thorlabs Vendas de Fotônicos Ltda.
Rua Rosalino Bellini, 175
Jardim Santa Paula
São Carlos, SP, 13564-050
Customer Support
- Phone: +55-16-3413 7062
- E-mail: brasil@thorlabs.com
デモルームのご案内
(クリックすると詳細がご覧いただけます)
日本 (東京都練馬区)
ソーラボジャパン株式会社
東京都練馬区北町3-6-3
お問い合わせ
- Tel: 03-6915-7701
- Email: techsupport.jp@thorlabs.com
デモルーム常設顕微鏡システム *ほかのデモをご希望の場合もご相談ください。
- 多光子顕微鏡Bergamo®IIシリーズ
- Cerna®顕微鏡(基本構成)
- OCTシステムTelesto®
- OCTシステムGanymede™(旧モデル)
Sterling, Virginia, USA
Thorlabs Imaging Systems HQ
108 Powers Court
Sterling, VA 20166
Customer Support
- Phone: (703) 651-1700
- E-mail: ImagingTechSupport@thorlabs.com
Demo Rooms
- Bergamo® II Series Multiphoton Microscopes
- Veneto® Inverted Microscopes
- Four-Channel Cerna®-Based Confocal Microscopes
- Cerna Birefringence Imaging Microscopes
- Multiphoton Mesoscope
- OCT Systems: Telesto® and Ganymede™
Lübeck, Germany
Thorlabs GmbH
Maria-Goeppert-Straße 9
23562 Lübeck
Customer Support
- Phone: +49 (0) 8131-5956-40840
- Email: oct@thorlabs.com
Demo Rooms
- Ganymede™ Series SD-OCT Systems
- Telesto® Series SD-OCT Systems
- Telesto® Series PS-OCT Systems
- Atria® Series SS-OCT Systems
- Vega™ Series SS-OCT Systems
Shanghai, China
Thorlabs China
Room A101, No. 100, Lane 2891, South Qilianshan Road
Shanghai 200331
Customer Support
- Phone: +86 (0)21-60561122
- Email: techsupport-cn@thorlabs.com
Demo Rooms
- Bergamo® II Series Multiphoton Microscopes
- Cerna Birefringence Imaging Microscopes
- OCT Systems: Telesto® and Ganymede™
| Posted Comments: | |
gaiqing Wang
(posted 2020-05-07 11:13:33.577) I am looking for a cheap way to do confocal imaging in vivo. Is this Bergamo II Series Multiphoton Microscope my best option? Can you send me a quote? YLohia
(posted 2020-05-07 09:45:11.0) Thank you for contacting Thorlabs. We will reach out to you directly to discuss your requirements. jfpena
(posted 2016-12-19 18:15:55.003) I am looking for a cheap way to do confocal imaging in vivo. Is this Bergamo II Series Multiphoton Microscope my best option? Can you send me a quote? tfrisch
(posted 2016-12-22 11:44:31.0) Hello, thank you for contacting Thorlabs. A member of our Imaging Team will reach out to you directly to discuss this system and your application. birech
(posted 2016-11-17 06:33:49.463) I asked for a price quote for this product, Bergamo II Series Multiphoton Microscopes three days ago. I am working at the University of Nairobi in Kenya and would wish to order one.
Regards,
Birech tfrisch
(posted 2016-11-17 06:56:23.0) Hello, thank you for contacting Thorlabs. I have forwarded this request to our Imaging Sales Team. I apologize for the delay. |
 Products Home
Products Home










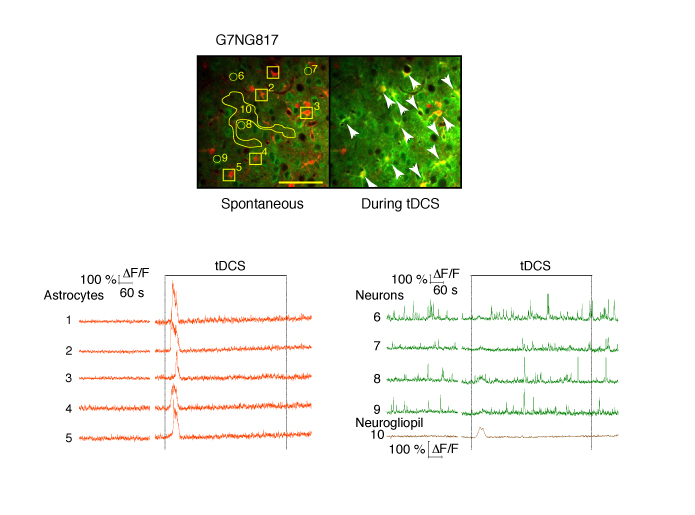

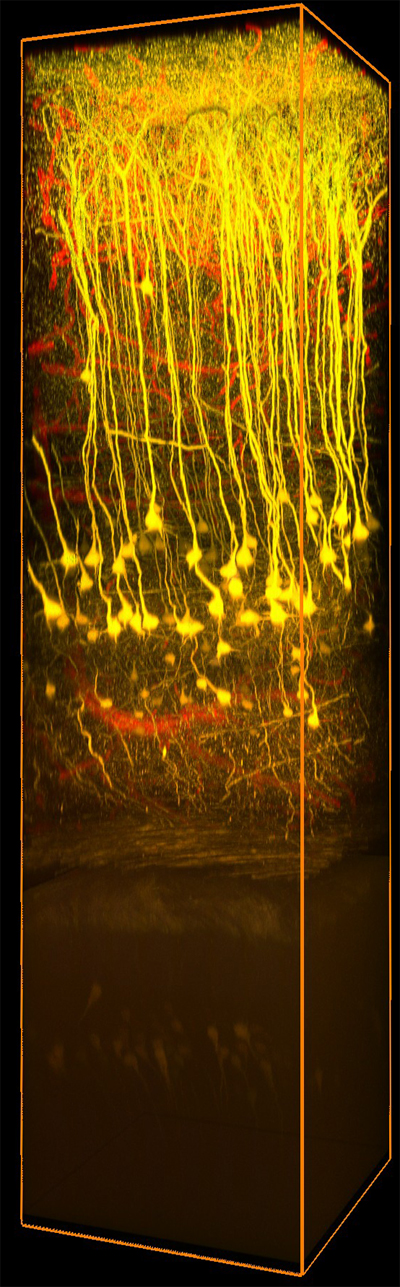
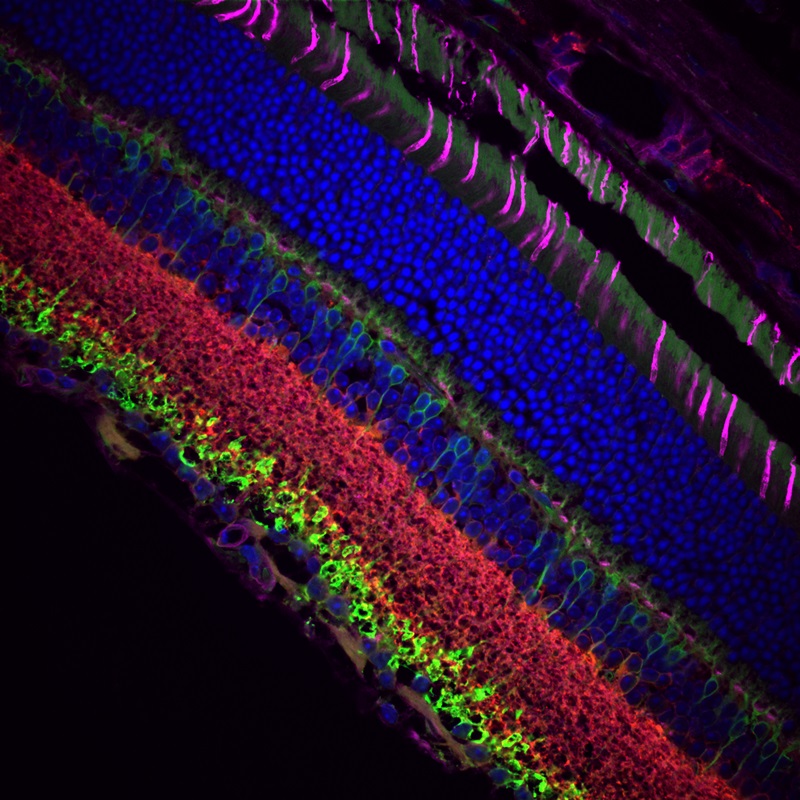
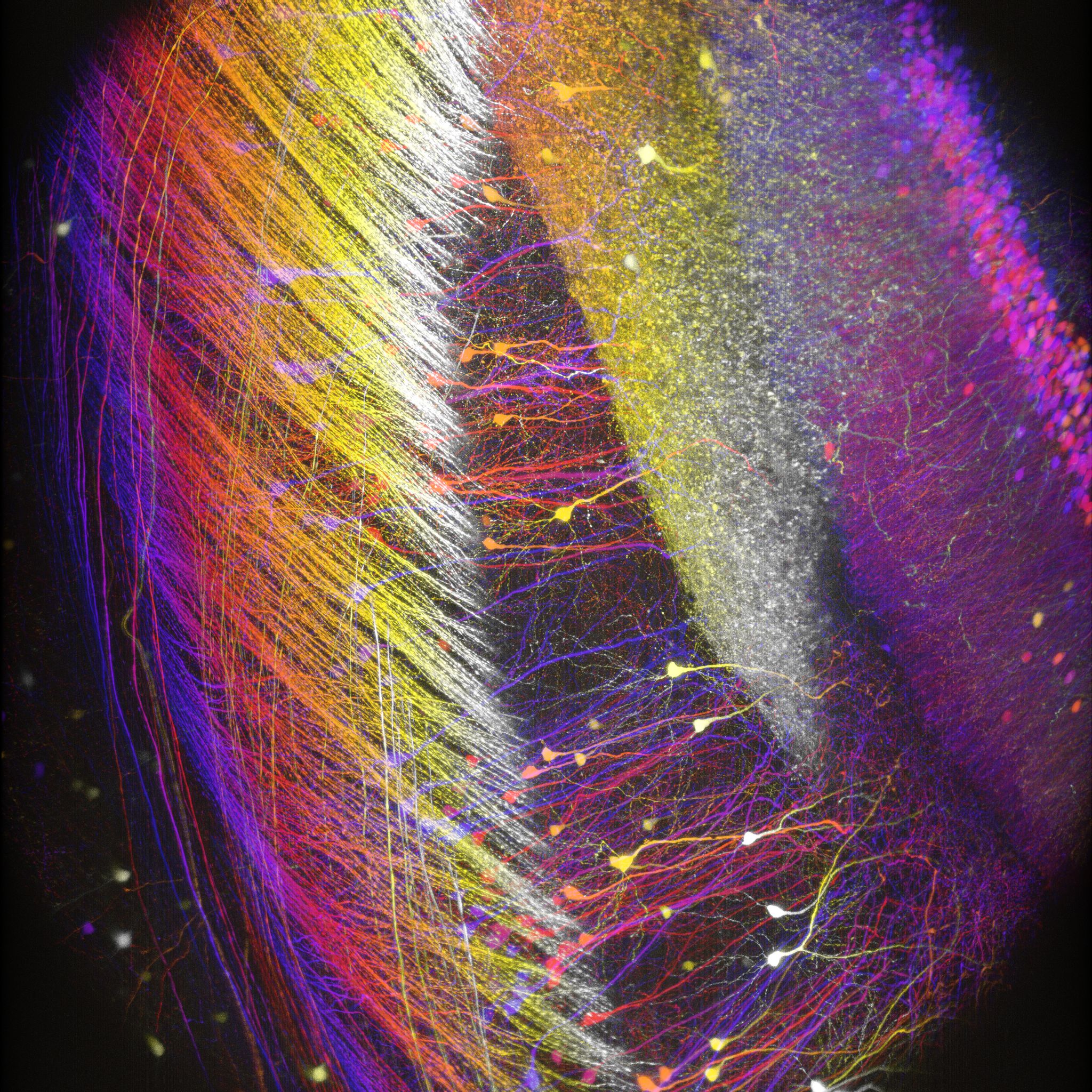
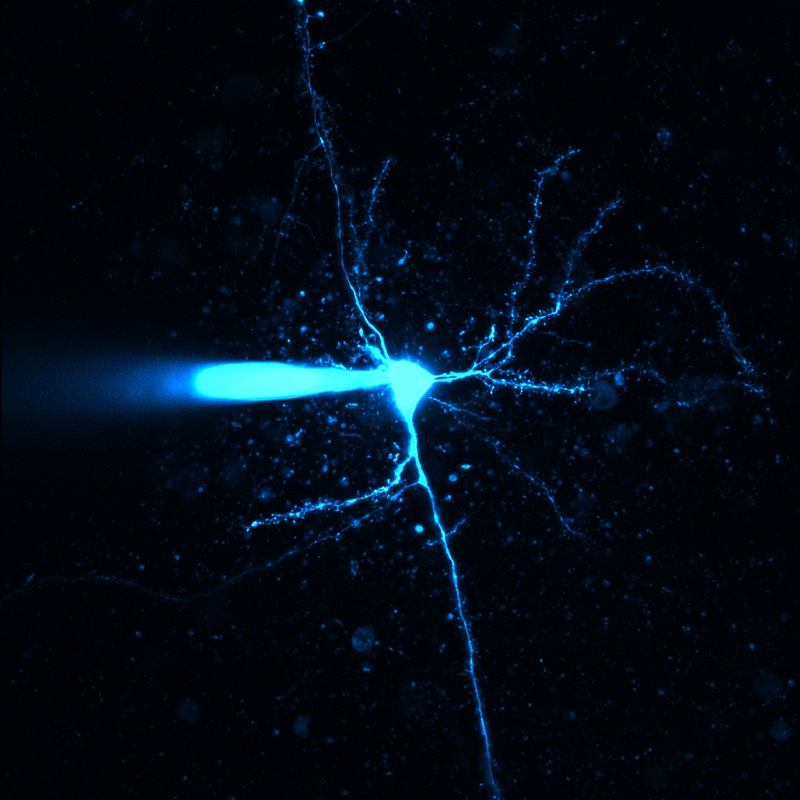
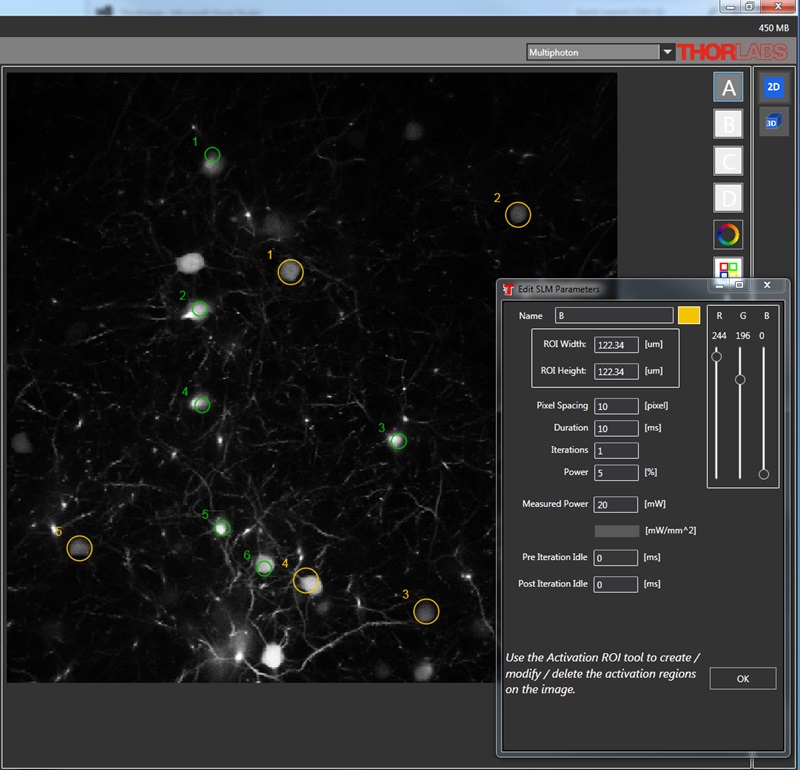
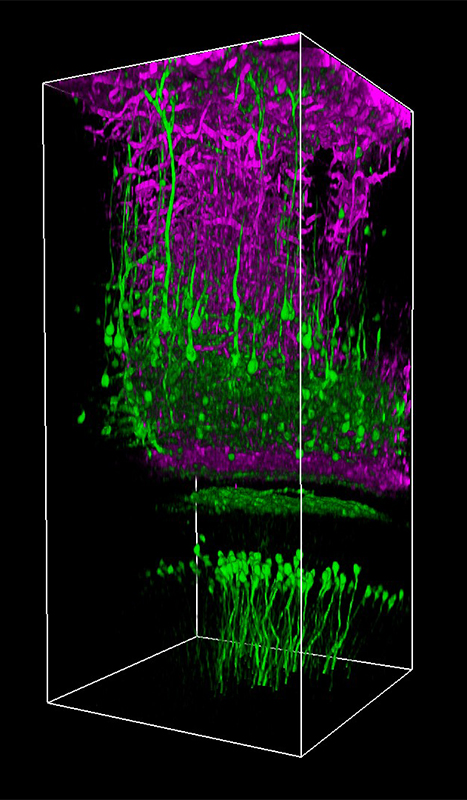
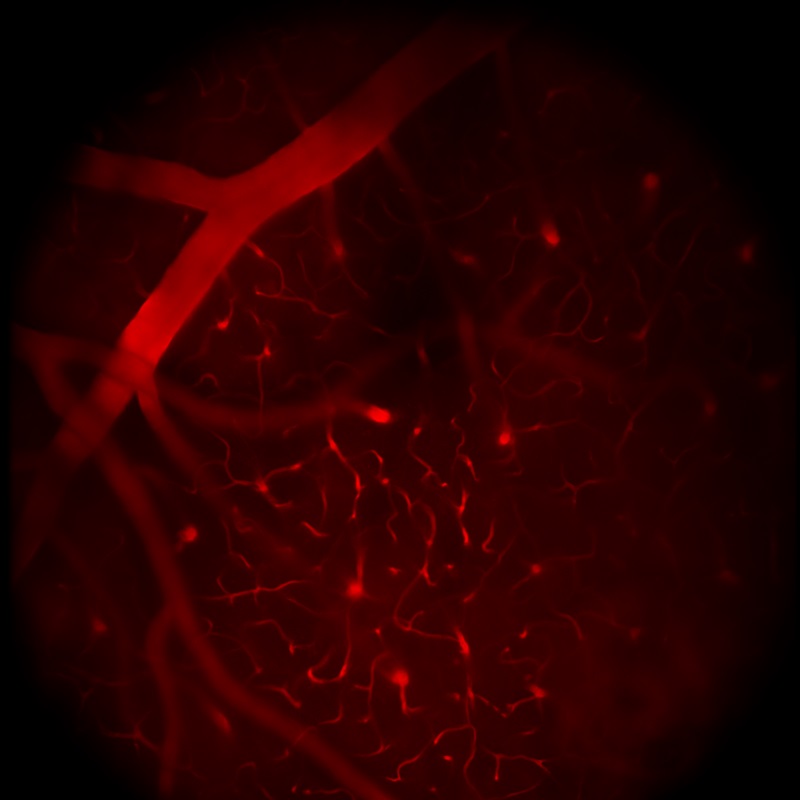
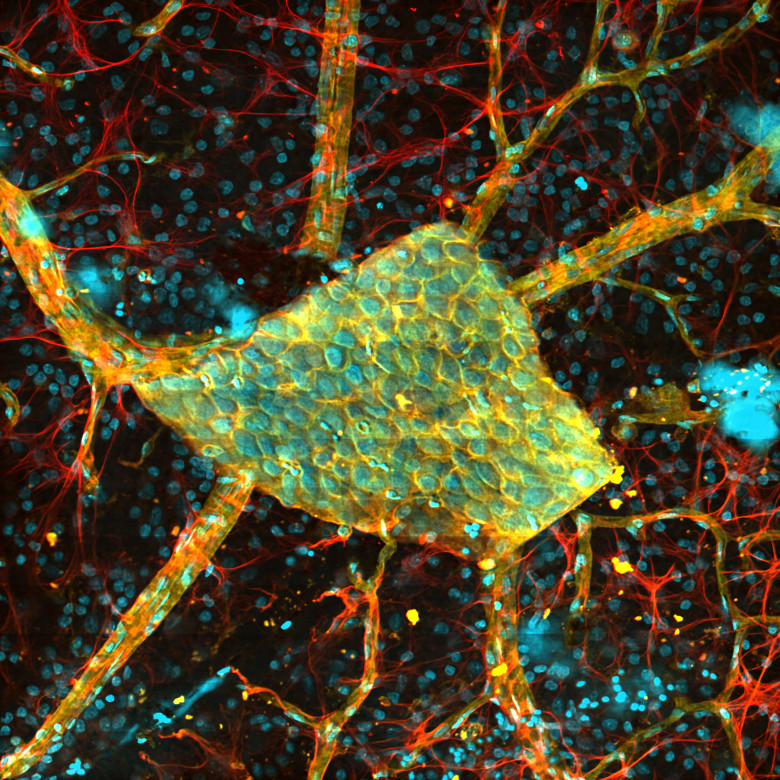

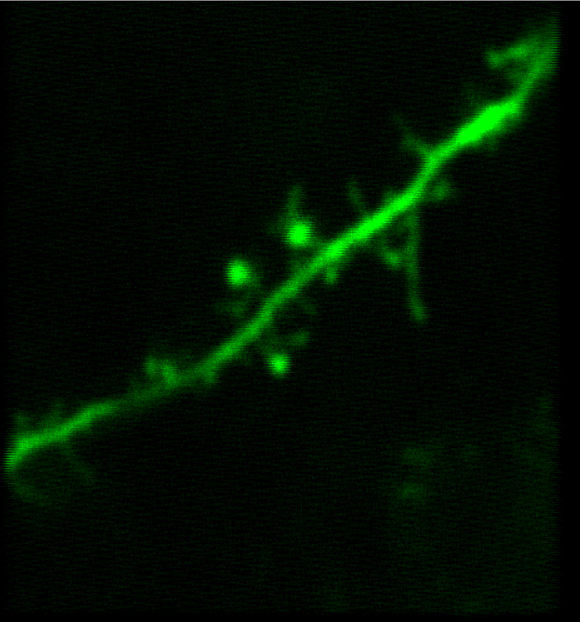
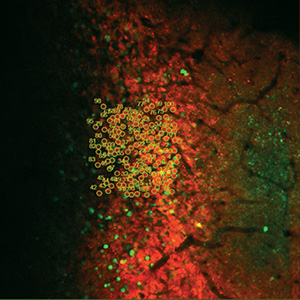
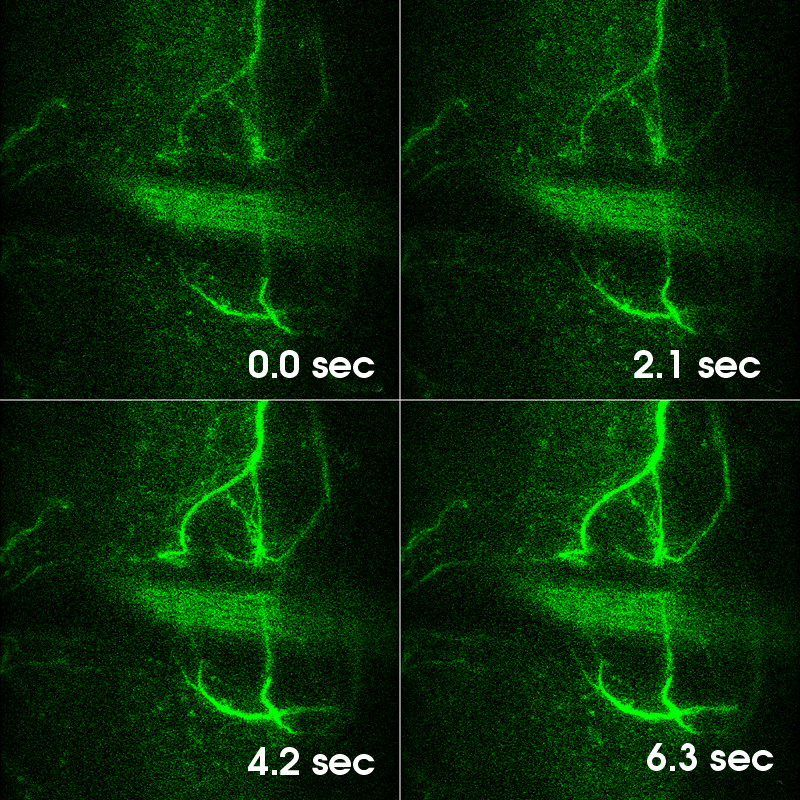












 顕微鏡Bergamo® IIIシリーズ
顕微鏡Bergamo® IIIシリーズ